1. The Chemical Properties of Acids and Bases
- Books Name
- Chemistry Class 10 NCERT based
- Publication
- Grow Career Publication
- Course
- CBSE Class 10
- Subject
- Chemistry
Indicators: Indicators are substances which indicate the acidic or basic nature of the solution by the colour change.
Types of Indicator: Some common types of indicators are:
1. Natural Indicators: Indicators obtained from natural sources are called Natural Indicators. Litmus, turmeric, red cabbage, China rose, etc., are some common natural indicators used widely to show the acidic or basic character of substances.
Turmeric: Turmeric is another natural indicator. Turmeric is yellow in colour. Turmeric solution or paper turns reddish brown with base. Turmeric does not change colour with acid.
Red Cabbage: The juice of red cabbage is originally purple in colour.
2. Olfactory Indicator: Substances which change their smell when mixed with acid or base are known as Olfactory Indicators. For example; Onion, vanilla etc.
Onion: Paste or juice of onion loses its smell when added with base. It does not change its smell with acid.
Vanilla: The smell of vanilla vanishes with base, but its smell does not vanish with an acid.
3. Synthetic Indicator: Indicators that are synthesized in the laboratory are known as Synthetic Indicators. For example; Phenolphthalein, methyl orange, etc.
Phenolphthalein is a colour less liquid. It remains colour less with acid but turns into pink with a base.
Methyl orange is originally orange in colour. It turns into the red with acid and turns into yellow with base.
Acids, Bases and Salts
Classification of matter
On the basis of
a) composition – elements, compounds and mixtures
b) state – solids, liquids and gases
c) solubility – suspensions, colloids and solutions
Types of mixtures – homogeneous and heterogeneous
Types of compounds – covalent and ionic
an Acid and a Base?
Ionisable and non-ionisable compounds
An ionisable compound when dissolved in water or in its molten state, dissociates into ions almost entirely. Example: NaCl, HCl, KOH, etc.
A non-ionisable compound does not dissociate into ions when dissolved in water or in its molten state. Example: glucose, acetone, etc.
Arrhenius theory of acids and bases
Acids: Acids are sour in taste, turn blue litmus red, and dissolve in water to release H+ ions.
Properties of Acids:
Acids have a sour taste.
Turns blue litmus red.
Acid solution conducts electricity.
Release H+ ions in aqueous solution.
Types of Acids: Acids are divided into two types on the basis of their occurrence i.e., Natural acids and Mineral acids.
(i) Natural Acids: Acids which are obtained from natural sources are called Natural Acids or Organic Acids.
Examples:
Methanoic acid (HCOOH)
Acetic acid (CH3COOH)
Oxalic acid (C2H2O4) etc.
(ii) Mineral Acids: Acids that are prepared from minerals are known as Mineral Acids Example; Inorganic acids, man-made acids or synthetic acid are also known as Mineral Acids.
Example:
Nitric acid (HNO3)
Carbonic acid (H2CO3)
Phosphoric acid (H3PO4) etc.
Chemical Properties of Acid:
(i) Reaction of acids with metal: Acids give hydrogen gas along with respective salt when they react with a metal.

Metal + Acid → Salt + Hydrogen
Test For Hydrogen Gas: The gas evolved after reaction of acid with metal can be tested by bringing a lighted candle near it. If the gas bums with a pop sound, then it confirms the evolution of hydrogen gas.
(ii) Reaction of acids with metal carbonate: Acids give carbon dioxide gas and respective salts along with water when they react with metal carbonates.
![]()
Metal carbonate + Acid → Salt + Carbon dioxide + Water

(iii) Reaction of acid with hydrogen carbonates (bicarbonates): Acids give carbon dioxide gas, respective salt and water when they react with metal hydrogen carbonate.

Example:
Test For Evolution of Carbon Dioxide Gas: Carbon dioxide turns lime water milky when passed through it. This is the characteristic test for carbon dioxide gas.
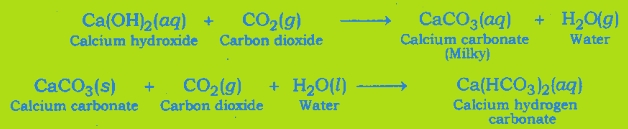
The gas evolved because of reaction of the acid with metal carbonate or metal hydrogen carbonate turns lime water milky. This shows that the gas is carbon dioxide gas.
Acids
- Strong Acids
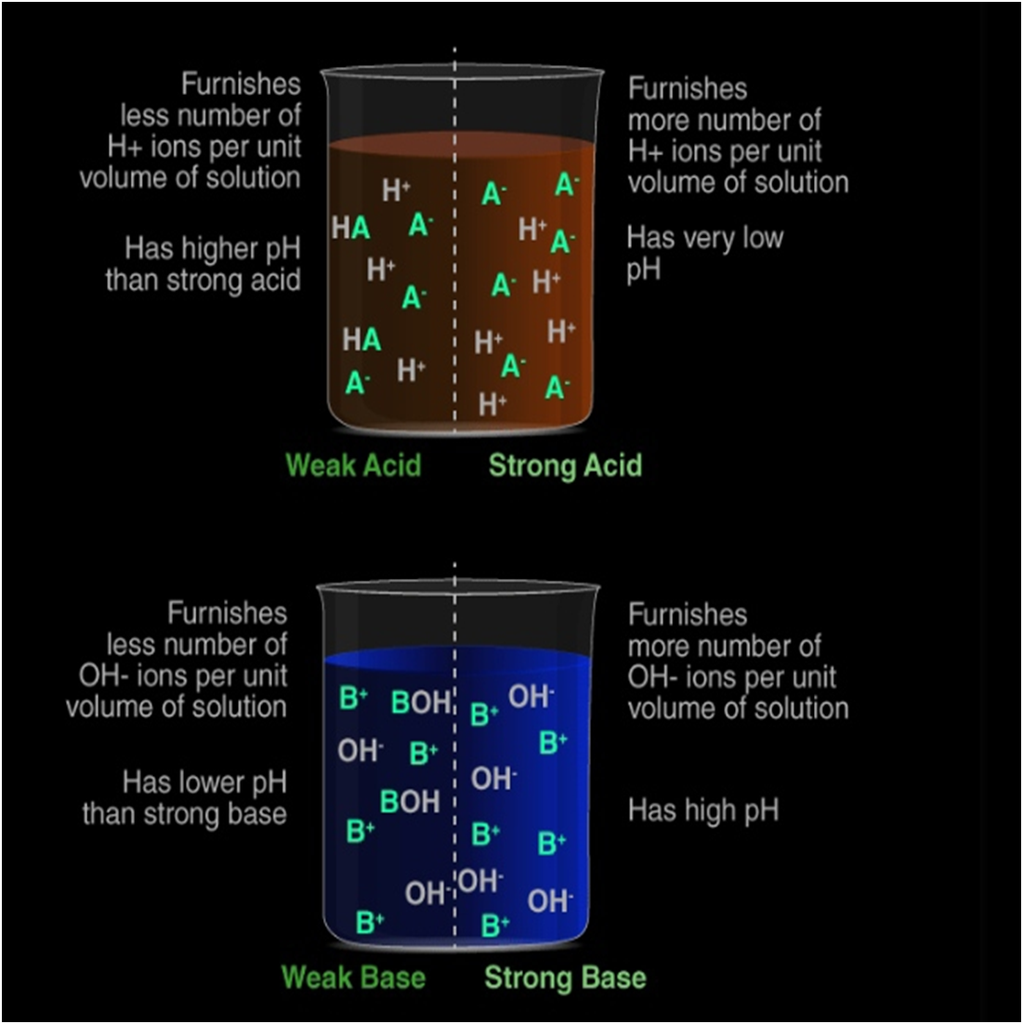
An acid which is completely ionised in water and produces (H+) is called Strong Acid.
Examples: Hydrochloric acid (HCl), Sulphuric acid (H2SO4), Nitric acid (HNO3)
- Weak Acids
An acid which is partially ionised in water and thus produces a small amount of hydrogen ions (H+) is called a Weak Acid.
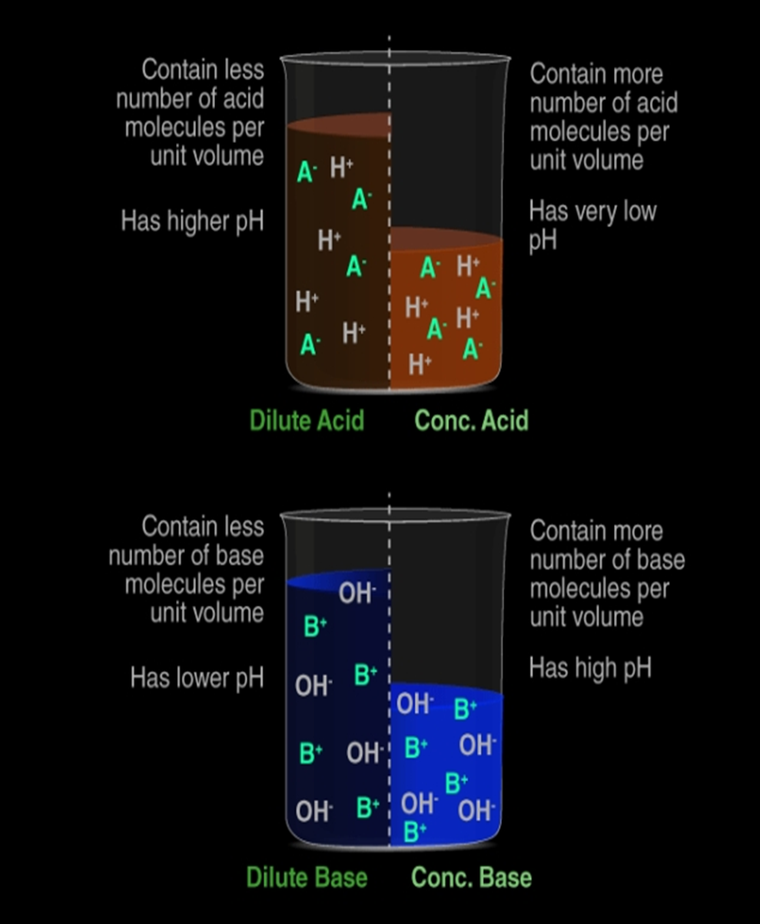
Example: Acetic acid (CH3COOH), Carbonic acid (H2CO3) , When a concentrated solution of acid is diluted by mixing water, then the concentration of Hydrogen ions (H+) or hydronium ion (H3O–) per unit volume decreases.
Bases: Bases are bitter in taste, have soapy touch, turn red litmus blue and give hydroxide ions (OH–) in aqueous solution.
Examples: Sodium hydroxide (caustic soda) – NaOH
Calcium hydroxide – Ca(OH)2
Potassium hydroxide (caustic potash) – (KOH)
Properties of Bases:
1.Have a bitter taste.
2.Turns red litmus blue.
3.Conducts electricity in solution.
4.Release OH– ions in Aqueous Solution
Types of bases: Bases can be divided in two types – Water soluble and Water-insoluble.
The hydroxide of alkali and alkaline earth metals are soluble in water. These are also known as alkali.
For example; sodium hydroxide, magnesium hydroxide, calcium hydroxide, etc.
Chemical properties of bases:
(i) Reaction of Base with Metals: When alkali (base) reacts with metal, it produces salt and hydrogen gas.
Alkali + Metal → Salt + Hydrogen
(ii) Reaction of Base with Oxides of Non-metals: Non-metal oxides are acidic in nature. For example; carbon dioxide is a non-metal oxide. When carbon dioxide is dissolved in water it produces carbonic acid.
Therefore, when a base reacts with non-metal oxide, both neutralize each other resulting respective salt and water.
![]()
Base + Non-metal oxide → Salt + Water
(iii) Neutralisation Reaction: An acid neutralizes a base when they react with each other and respective salt and water are formed.
![]()
Acid + Base → Salt + Water
(iv) Reaction of Acid with Metal Oxides: Metal oxides are basic in nature. Thus, when an acid reacts with a metal oxide both neutralize each other. In this reaction, the respective salt and water are formed.
![]()
Acid + Metal Oxide → Salt + Water
Common in all bases: A base dissociates hydroxide ion in water, which is responsible for the basic behaviour of a compound.
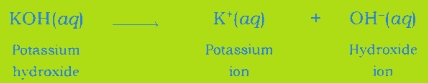
Example: When sodium hydroxide is dissolved in water, it dissociates hydroxide ion and sodium ion.
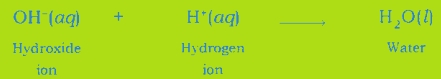
Neutralisation Reaction: When an acid reacts with a base, the hydrogen ion of acid combines with the hydroxide ion of base and forms water
Dilution of Acid and Base: The hydrogen ion in an acid and hydroxide ion in a base, per unit volume, shows the concentration of acid or base.By mixing of acid to water, the concentration of hydrogen ion per unit volume decreases. Similarly, by addition of base to water, the concentration of hydroxide ion per unit volume decreases. This process of addition of acid or base to water is called Dilution and the acid or base is called Diluted.The dilution of acid or base is exothermic. Thus, acid or base is always added to water and water is never added to acid or base. If water is added to a concentrated acid or base, a lot of heat is generated, which may cause splashing out of acid or base and may cause severe damage as concentrated acid and base are highly corrosive
Examples
Acids
1.Hydrochloric acid (HCl)
2.Sulphuric acid (H2SO4)
3.Nitric acid (HNO3)
Bases
1.Sodium hydroxide (NaOH)
2.Potassium hydroxide (KOH)
3.Calcium hydroxide (Ca(OH)2)
Bronsted Lowry theory
1.A Bronsted acid is an H+ (aq) ion donor.
2.A Bronsted base is an H+ (aq) ion acceptor.
Example
In the reaction: HCl (aq) + NH3 (aq) → NH+4(aq) + Cl− (aq)
HCl – Bronsted acid and Cl− : its conjugate acid
NH3 – Bronsted base and NH+4 : its conjugate acid
Physical test
Given are two possible physical tests to identify an acid or a base.
a. Taste
An acid tastes sour whereas a base tastes bitter.
The method of taste is not advised as an acid or a base could be contaminated or corrosive.
b. Effect on indicators by acids and bases
An indicator is a chemical substance which shows a change in its physical properties, mainly colour or odour when brought in contact with an acid or a base.
Below mentioned are commonly used indicators and the different colours they exhibit:
a) Litmus
In a neutral solution – purple
In acidic solution – red
In basic solution – blue
b) Methyl orange
In a neutral solution – orange
In acidic solution – red
In basic solution – yellow
c) Phenolphthalein
In a neutral solution – colorless
In acidic solution – remains colorless
In basic solution – pink
Acid-Base Reactions & Reactions of acids and bases
a) Reaction of acids and bases with metals
Acid + active metal → salt + hydrogen + heat
2HCl + Mg → MgCl2 + H2 (↑)
2NaOH + Zn → Na2ZnO2 + H2 (↑)
b) Reaction of acids with metal carbonates and bicarbonates
Acid + metal carbonate or bicarbonate → salt + water + carbon dioxide.
2HCl + CaCO3 → CaCl2 + H2O + CO2
H2SO4 + Mg (HCO3)2 → MgSO4 + 2H2O + 2CO2
c) Neutralisation reaction
1. Reaction of metal oxides and hydroxides with acids
Metal oxides or metal hydroxides are basic in nature.
Acid + base → salt + water + heat
H2SO4 + MgO → MgSO4 + H2O
2HCl + Mg (OH) 2 → MgCl2 + 2H2O
2. Reaction of non-metal oxides with bases
Non-metal oxides are acidic in nature
Base + Nonmetal oxide → salt + water + heat
2NaOH + CO2→ Na2CO3 + H2O
Base:
Bases undergo neutralisation reaction with acids.
They are comprised of metal oxides, metal hydroxides, metal carbonates and metal bicarbonates.
Most of them are insoluble in water.
2. Common Things in All Acids and Bases
- Books Name
- Chemistry Class 10 NCERT based
- Publication
- Grow Career Publication
- Course
- CBSE Class 10
- Subject
- Chemistry
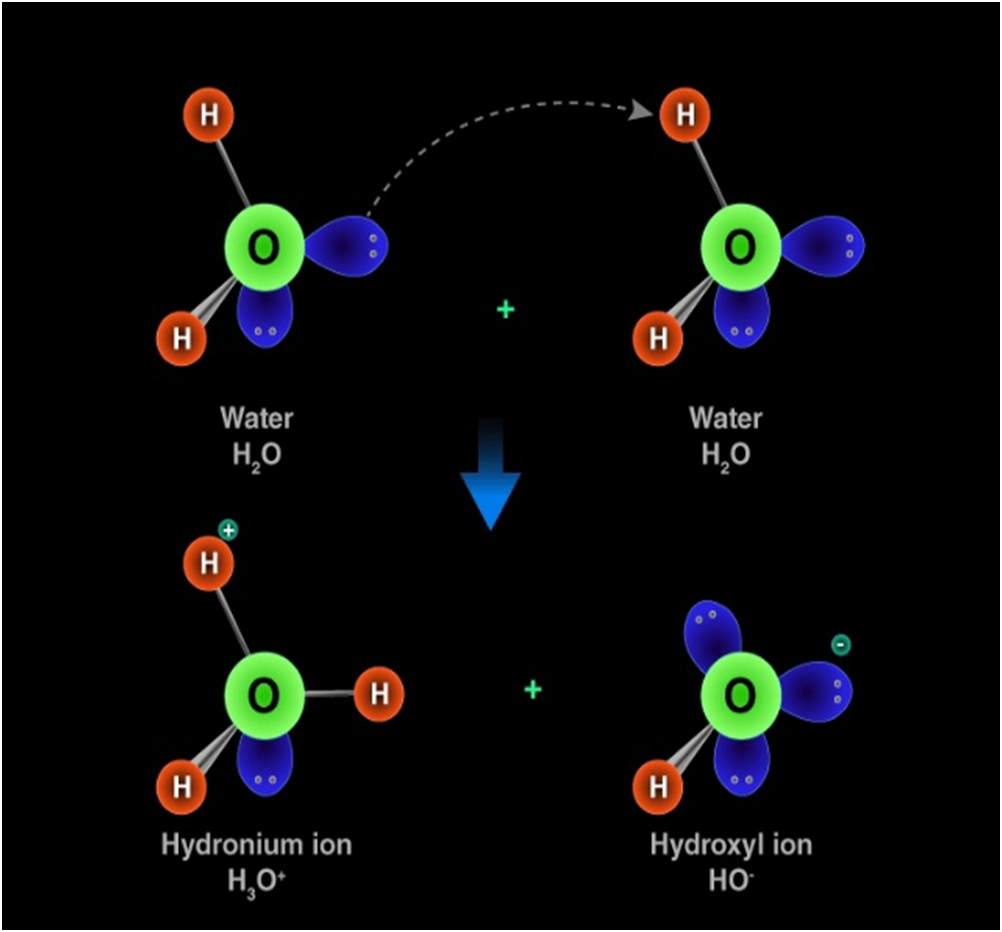
Hydronium ion
Hydronium ion is formed when a hydrogen ion accepts a lone pair of electrons from the oxygen atom of a water molecule, forming a coordinate covalent bond.
Dilution
Dilution is the process of reducing the concentration of a solution by adding more solvent (usually water) to it.
It is a highly exothermic process. Strong acid or base: When all molecules of a given amount of an acid or a base dissociate completely in water to furnish their respective ions, H+(aq) for acid and OH−(aq) for base).
Weak acid or base: When only a few of the molecules of a given amount of an acid or a base dissociate in water to furnish their respective ions, H+(aq) for acid and OH−(aq) for base).
Dilute acid: contains less number of H+(aq) ions per unit volume.
Concentrated acid: contains more number of H+(aq) ions per unit volume
3. Strongness about Acids or Bases
- Books Name
- Chemistry Class 10 NCERT based
- Publication
- Grow Career Publication
- Course
- CBSE Class 10
- Subject
- Chemistry
Universal indicator
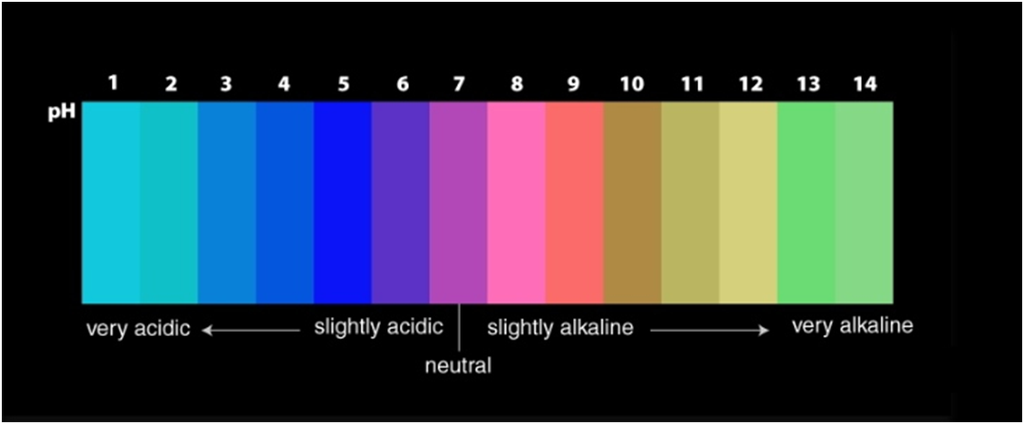
A universal indicator has a pH range from 0 to 14 that indicates the acidity or alkalinity of a solution.
A neutral solution has pH=7
pH
pH=−log10[H+]
In pure water, [H+]=[OH−]=10−7 mol/L. Hence, the pH of pure water is 7.
The pH scale ranges from 0 to 14.
If pH < 7 → acidic solution
If pH > 7→ basic solution
Importance of pH in everyday life
1. pH sensitivity of plants and animals
Plants and animals are sensitive to pH. Crucial life processes such as digestion of food, functions of enzymes and hormones happen at a certain pH value.
2. pH of a soil
The pH of a soil optimal for the growth of plants or crops is 6.5 to 7.0.
3. pH in the digestive system
The process of digestion happens at a specific pH in our stomach which is 1.5 to 4.
The pH of the interaction of enzymes, while food is being digested, is influenced by HCl in our stomach.
4. pH in tooth decay
Tooth decay happens when the teeth are exposed to an acidic environment of pH 5.5 and below.
5. pH of self-defence by animals and plants
Acidic substances are used by animals and plants as a self-defence mechanism. For example, bee and plants like nettle secrete a highly acidic substance for self-defence. These secreted acidic substances have a specific pH.
4. About Salts
- Books Name
- Chemistry Class 10 NCERT based
- Publication
- Grow Career Publication
- Course
- CBSE Class 10
- Subject
- Chemistry
Salts
A salt is a combination of an anion of an acid and a cation of a base.
Examples – KCl, NaNO3 ,CaSO4, etc.
Salts are usually prepared by the neutralisation reaction of an acid and a base.
Common salt
Sodium Chloride (NaCl) is referred to as common salt because it’s used all over the world for cooking. Sodium chloride (NaCl) is also known as Common or Table Salt. It is formed after the reaction between sodium hydroxide and hydrochloric acid. It is a neutral salt. The pH value of sodium chloride is about 7. Sodium chloride is used to enhance the taste of food. Sodium chloride is used in the manufacturing of many chemicals.
Family of salts
Salts having the same cation or anion belong to the same family. For example, NaCl, KCl, LiCl.
pH of salts
A salt of a strong acid and a strong base will be neutral in nature. pH = 7 (approx.).
A salt of a weak acid and a strong base will be basic in nature. pH > 7.
A salt of a strong acid and a weak base will be acidic in nature. pH < 7.
The pH of a salt of a weak acid and a weak base is determined by conducting a pH test.
Preparation of Sodium hydroxide
Chemical formula – NaOH
Also known as – caustic soda
Preparation (Chlor-alkali process):
Electrolysis of brine (solution of common salt, NaCl) is carried out.
At anode: Cl2 is released
At cathode: H2 is released
Sodium hydroxide remains in the solution.
Bleaching powder
Chemical formula – Ca(OCl)Cl or CaOCl2
Preparation – Ca(OH)2(aq)+Cl2(g)→CaOCl2(aq)+H2O(l)
On interaction with water – bleaching powder releases chlorine which is responsible for bleaching action.
Baking soda
Chemical name – Sodium hydrogen carbonate
Chemical formula – NaHCO3
Preparation (Solvay process):
a. Limestone is heated: CaCO3→CaO+CO2
b. CO2 is passed through a concentrated solution of sodium chloride and ammonia:
NaCl(aq)+NH3(g)+CO2(g)+H2O(l)→NaHCO3(aq)+NH4Cl(aq)
Uses:
1. Textile industry
2. Paper industry
3. Disinfectant
Washing soda
Chemical name – Sodium hydrogen carbonate
Chemical formula – NaHCO3
Preparation (Solvay process) –
a. Limestone is heated: CaCO3 → CaO + CO2
b. CO2 is passed through a concentrated solution of sodium chloride and ammonia:
NaCl(aq) + NH3(g) + CO2(g) + H2O(l) → NaHCO3(aq) + NH4Cl(aq)
Uses
1. In glass, soap and paper industries
2. Softening of water
3. Domestic cleaner
Crystals of salts
Certain salts form crystals by combining with a definite proportion of water. The water that combines with the salt is called water of crystallisation.
Plaster of paris
Gypsum, CaSO4.2H2O (s) on heating at 100°C (373K) gives CaSO4. ½ H2O and 3/2 H2O
CaSO4. ½ H2O is plaster of paris.
CaSO4. ½ H2O means two formula units of CaSO4 share one molecule of water.
Uses – cast for healing fractures.

 PRIDE LEARNING PUBLICATION
PRIDE LEARNING PUBLICATION
 Grow Career Publication
Grow Career Publication
