- Books Name
- ACME SMART COACHING Chemistry Book
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Chemistry
Aryl Halides
Preparation of Aryl Halides
1. By Halogenation :
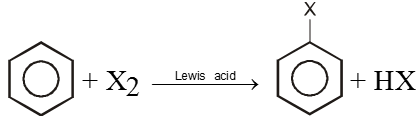
X2 = Cl2, Br2 ; Lewis acid = FeCl3, AlCl3, ZnCl2, etc.
2. By Decarboxylation :
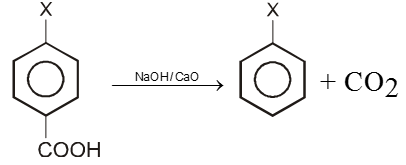
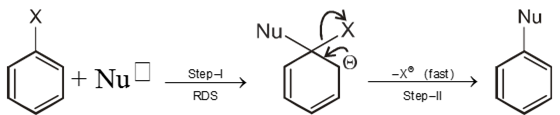
Chemical reaction of Aryl halide (Bimolecular nucleophilic substitution SN2 Ar)
- An electron withdrawing group at ortho or para positions with respect to a good leaving groups are necessary conditions for SN2 Ar.
Mechanism :
Intermediate ion is stabilized by resonance and are stable salts called Meisenheimer salts.
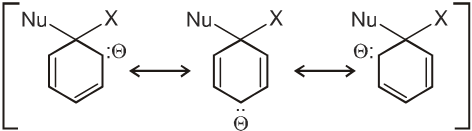
- A group that withdraws electrons tends to neutralize the negative charge of the ring and this dispersal of the charge stabilizes the carbanion.
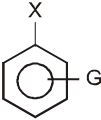
G withdraws electrons : stabilizes carbanion, activates the Ar-SN2 reaction.
(–![]() (CH3)3, –NO2, –CN, –SO3H, –COOH, –CHO, –COR, –X)
(CH3)3, –NO2, –CN, –SO3H, –COOH, –CHO, –COR, –X)
- A group that releases electrons tends to intensify the negative charge, destabilizes the carbanion, and thus slows down reaction.
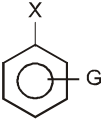
G (–NH2, –OH, –OR, –R) releases electrons : destabilizes carbanion, deactivates the Ar-SN2 reaction.
Element effect :- The fact that fluoro is the best leaving group among the halogens in most ArSN2 but in SN1 & SN2 mechanism where fluoro is the poorest leaving group among halogens.

 ACME SMART PUBLICATION
ACME SMART PUBLICATION
