- Books Name
- Ritan Sheth Chemistry Book
- Publication
- Ritan Sheth
- Course
- CBSE Class 11
- Subject
- Chemistry
PERCENTAGE COMPOSITION
One can check the purity of a given sample by analysing this data. Let us understand by taking the example of water (H20). Since water contains hydrogen and oxygen, the percentage composition of both these elements can be calculated as follows:
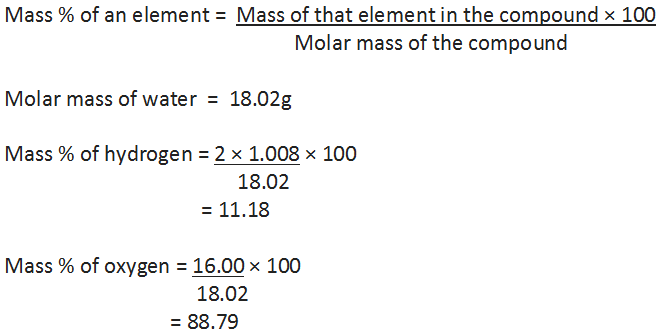
• Empirical Formula
The formula of the compound which gives the simplest whole number ratio of the atoms of yarious elements present in one molecule of the compound.
For example, the formula of hydrogen peroxide is H202. In order to express its empirical formula, we have to take out a common factor 2. The simplest whole number ratio of the atoms is 1:1 and the empirical formula is HO. Similarly, the formula of glucose is C6H1206. In order to get the simplest whole number of the atoms,
Common factor = 6
The ratio is = 1 : 2 : 1 The empirical formula of glucose = CH20
• Molecular Formula
The formula of a compound which gives the actual ratio of the atoms of various elements present in one molecule of the compound.
For example, molecular formula of hydrogen peroxide = H202and Glucose = C6H1206
Molecular formula = n x Empirical formula
Where n is the common factor and also called multiplying factor. The value of n may be 1, 2, 3, 4, 5, 6 etc.
In case n is 1, Molecular formula of a compound = Empirical formula of the compound.

 Ritan Sheth
Ritan Sheth
