structure of flower
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
FLOWER –
A Fascinating Organ of Angiosperms
Flowers are objects of aesthetic, ornamental, social, religious and cultural value. To a biologist, a flower is a modified condensed shoot to perform sexual reproduction in angiosperms.
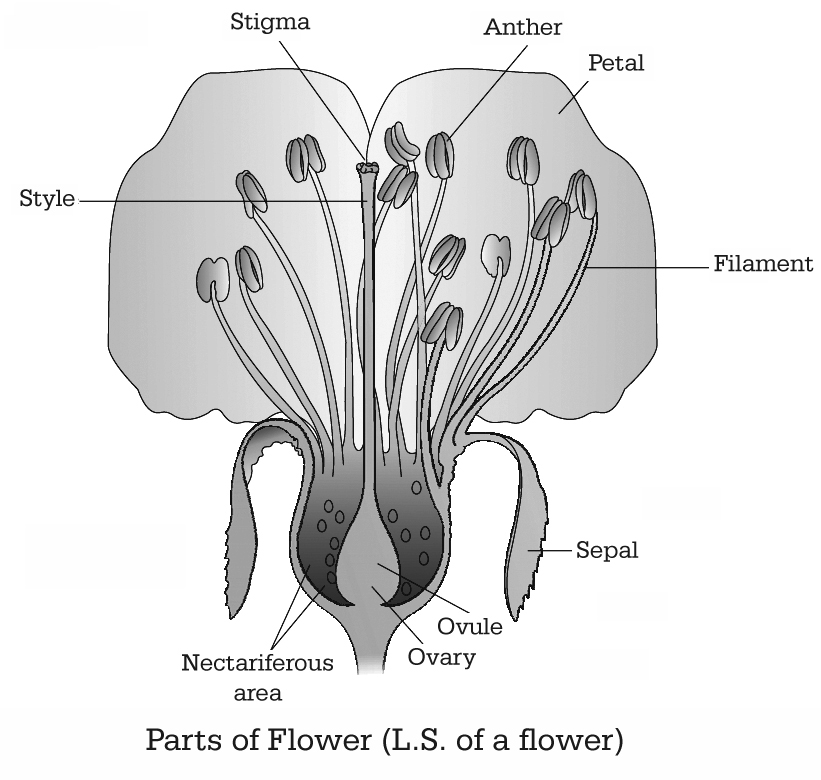
A typical angiospermic flower consists of four whorls of floral appendages attached on the receptacle - Calyx, Corolla, Androecium and Gynoecium.
Pre fertilisation: Structure and Events
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
PRE-FERTILIZATION : Structures and Events
Much before the actual flower is seen on a plant, the decision that the plant is going to flower has taken place. A number of hormonal and structural transformations occur prior to initiation of flowering.
Shoot apical meristem is transformed into reproductive meristem.
Reproductive meristem grows to form inflorescence axis over which floral primordia develop.
The primordia grow into floral buds and then flowers. In the flower, the androecium and gynoecium differentiate and develop.
Stamen, Microsporangium and Pollen grain
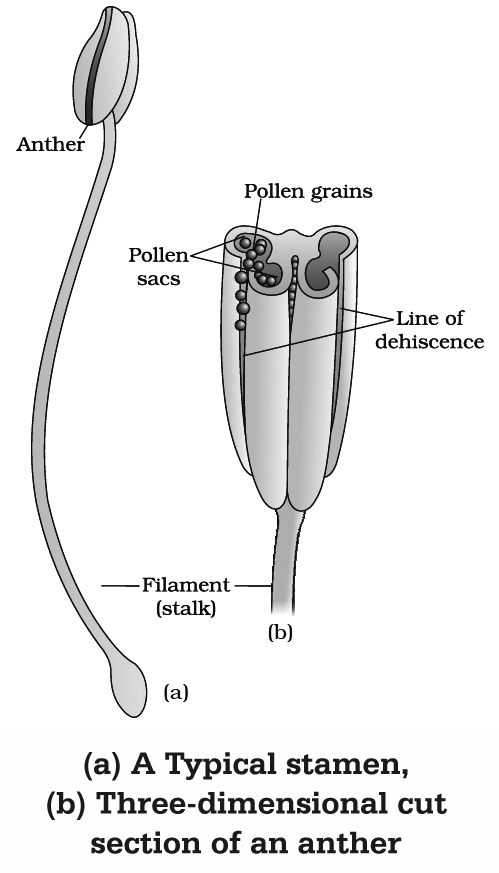
(a) Stamen or Microsporophyll (Male sex organ)
It consists of two parts :
(i) Anther
It is broader knob-like fertile part.
A typical angiospermic anther has two lobes and such anther is called dithecous.
Dithecous anther is a four sided (tetragonal) structure consisting of four microsporangia located at the corners, two in each lobe.
(ii) Filament
It is sterile, long and slender stalk.
The proximal end of the filament is attached to the thalamus, petal or tepal.
Concept Builder
(i) Floriculture is the science of cultivation, breeding, marketing and arrangement of flowers.
(ii) In members of Malvaceae, anther consists of one lobe and two microsporangia. Such anthers are monothecous and bisporangiate.
(iii) In Arceuthobium, the smallest dicot parasite, anther consists of one microsporangium, i.e., monothecous and monosporangiate
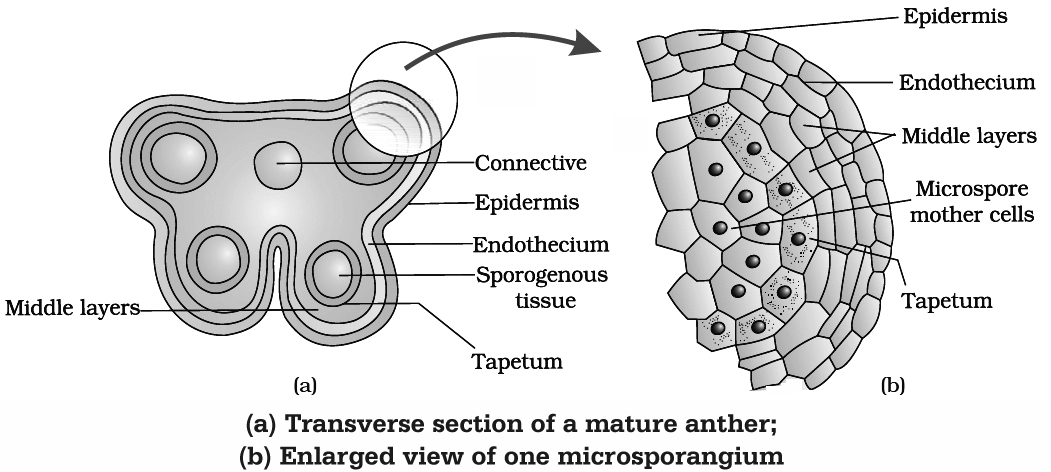
(b) Structure (T.S.) of Anther
A young anther consists of homogenous mass of meristematic cells called primary sporogenous cells surrounded by anther wall. Primary sporogenous cells form microspore mother cells (2n) inside the microsporangium.
Anther Wall Layers : Anther wall consists of following layers :
(1) Epidermis : Outermost single layered and protective in function. The epidermis of Arceuthobium develops some fibrous thickenings and is called exothecium.
(2) Endothecium : Cells of this layer have -cellulosic fibrous bands arising from inner tangential wall which help in dehiscence of anther due to their hygroscopic nature. Fibrous bands are absent in hydrophytes, e.g., Hydrocharitaceae.
(3) Middle layer : Cells of this layer are ephemeral and are 1-3 layered. It degenerates at maturity.
(4) Tapetum : This is the innermost layer of anther wall which surrounds the sporogenous tissue. Tapetal cells nourishes the developing pollen grains. Cells of the tapetum possess dense cytoplasm and generally have more than one nucleus. They are polyploid. The tapetal cells show increase in their DNA content.
Concept Builder
Increase in DNA content of tapetum may be 'achieved by the following ways:
(i) Endomitosis : It involves DNA replication and splitting of chromosomes through endoprophase, endometaphase, endoanaphase and endotelophase.
(ii) Formation of restitution nuclei : It involves normal mitosis upto anaphase but the chromosomes at two poles get surrounded by a common nuclear membrane so as to form a restitution nucleus.
(iii) Polyteny : If DNA replication is not accompanied by splitting of chromosomes, polytenic chromosomes are formed.
The tapetum is of two types :
(a) Secretory or glandular tapetum : These cells secrete sporopollenin, pollenkitt and compatibility proteins. These cells provide Ubisch bodies which help in the ornamentation of exine, as they have a chemical called sporopollenin which is deposited on them.
(b) Amoeboid or plasmodial or invasive tapetum : Cells undergo breakdown and their entire protoplasts move in the centre to nourish microspores.
(c) Microsporogenesis
The process of formation of microspores from a pollen mother cell (PMC) or microspore mother cell (MMC) through meiosis is called microsporogenesis. As each cell of the sporogenous tissue is capable of giving rise to a microspore tetrad, therefore each one is a potential pollen mother cell.
Cytokinesis, after the meiotic divisions in PMCs, is of two types:
(i) Successive : In this type, cytokinesis occurs after each meiotic division, thus isobilateral tetrad of microspores is formed, e.g., monocots. Successive type of cytokinesis is advanced type.
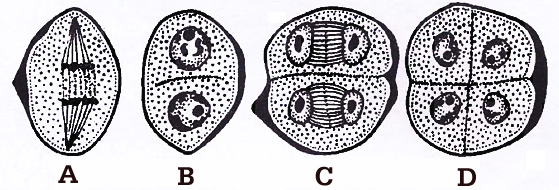
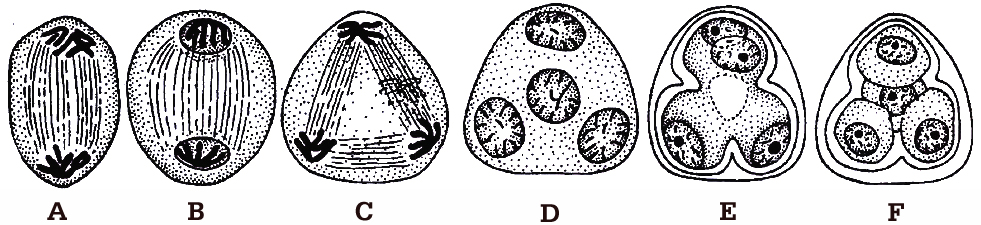
during microsporogenesis, (A-B) Dividing mother cell, (C) Dividing dyad, (D) Tetrad
(ii) Simultaneous : It occurs after complete meiotic (I and II) division, thus tetrahedral tetrad of microspores is formed, e.g., dicots.
The microspores, as they are formed, are arranged in a cluster of four cells, called microspore tetrad. Usually the arrangement of microspores in a tetrad is tetrahedral or isobilateral.
However, T-shaped, linear and decussate tetrads are also found. In Aristolochia elagans, all the five types of tetrads are present.
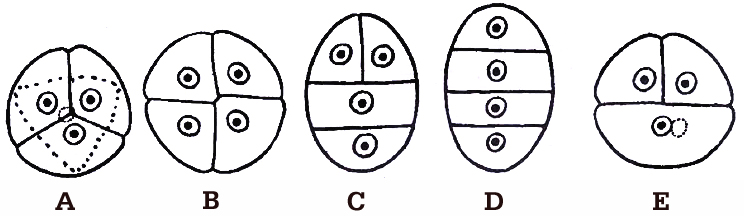
A. Tetrahedral, B. Isobilateral, C. T-shaped, D. Linear, E. Decussate
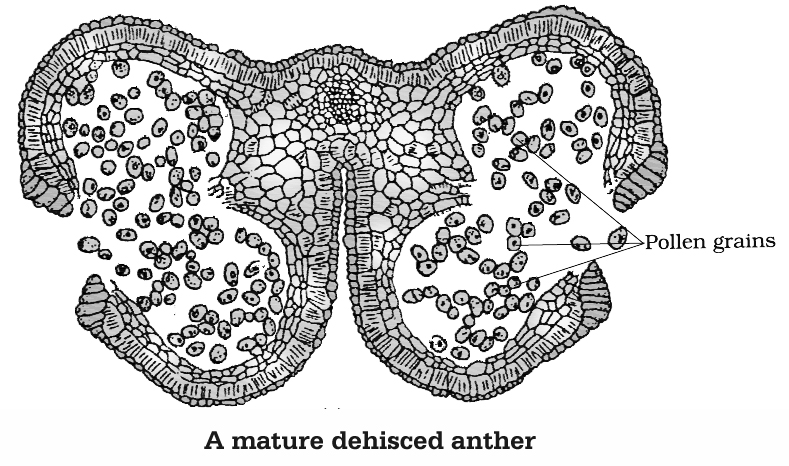
As the anthers mature and dehydrate, the microspores dissociate from each other and develop into pollen grains. These are released with the dehiscence of anther.
Concept Builder
(i) R. Camerarius described sexual reproduction for the first time in plants.
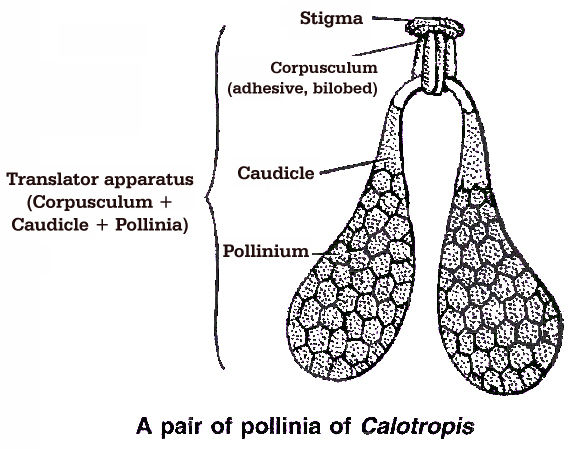
(ii) 'In family Asclepiadaceae (Calotropis) and Orchidaceae, all the microspores in a sporangium adhere together in a single mass known as pollinium.
(iii) In Calotropis, the pollinia of adjacent anthers of different stamens are attached by thread like caudicles (retinaculi) to a sticky disc called corpusculum. The whole structure is called translator.
(iv) After their formation, the microspores are separated from tetrad, but in Elodea, Drosera, Typha, the microspores do not separate from each other, thus developing into compound pollen grains.
(v) In family Cyperaceae, out of 4 microspores formed, 3 degenerate, so ultimately one MMC (2n) produces only one microspore or pollen grain.
(vi) Sometimes more than four microspores are produced from one microspore mother cell. It is called polyspory, e.g., Cuscuta reflexa.
(d) Pollen Grain
(i) These are generally spherical measuring about 25-50 µm in diameter. The cell wall of pollen grain is called sporoderm which consists of two layers. The hard outer layer called the exine and inner thin layer is called the intine.
(ii) Intine : It is made of cellulose and pectin.
(iii) Exine : The exine is made up of sporopollenin is a fatty substance and one of the most resistant organic material known. It is not affected by high temperature, strong acids or alkali. No enzyme is known to degrade it. Because of the presence of sporopollenin, pollen grains of the past plants are well preserved as fossils. Exine is made of two layers :
(a) Ektexine : It is highly sculptured and is differentiated into outer tectum, middle baculum and innermost foot layer. Tectum provides a characteristic sculpturing or designs over the surface of pollen grain. The design helps the experts to identify the pollen grain as to its class, family, genus or species.
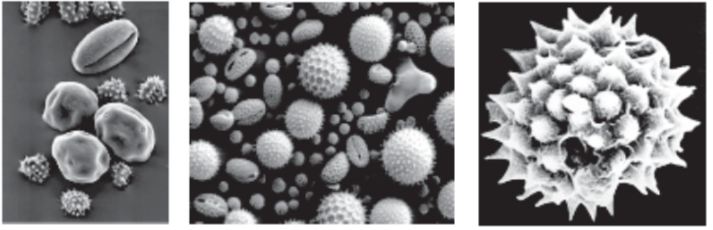
(b) Endexine: It is not sculptured.
(iv) Pollen grain exine has prominent apertures called germ pores where sporopollenin is absent. Pollen grains can be monocolpate (having one germ pore called germinal furrow, e.g., monocots), bicolpate (2 germ pores) and tricolpate (3 germ pores, e.g., dicots).
(v) The pollen kitt is a sticky layer found on the outer side of exine of mature pollen grains of many insect pollinated species, it is made of carotenoids and lipids. Pollenkitt material is contributed by the tapetal cells. Pollen kitt acts as an insect attractant and can help against UV.
(vi) Pollen grains of many species (especially anemophilous plants) cause severe allergies and bronchial affictions in some people. Weed Parthenium hysterophorus (carrot grass) came to India as a contaminant with improved wheat. The weed has become a major cause of pollen allergy. Hay fever is an allergic reaction due to the presence of pollen in the air. Plants commonly causing hay fever are Amaranthus, Chenopodium and Parthenium.
(vii) Pollen grains are rich in nutrients. They are taken as tablets and syrups to improve health. Pollen consumption has been claimed to enhance the performance of athletes and race horses.
(viii) The period for which pollen grains retain the ability to germinate is called Pollen Viability. It is highly variable and to some extent depends upon the environmental factors like temperature, humidity.
In cereals, like rice, wheat etc, pollen viability is minimum upto 30 minutes, while in Rosaceae, Leguminosae and Solanaceae it is upto several months.
(ix) Pollen grains can be cryopreserved in liquid nitrogen (-196°C) and used as pollen banks.
Concept Builder
(i) The study of pollen grain is called Palynology. (Term given by Hyde and Williams).
(ii) In Hyacinthus, Nemec observed 8-nucleated embryo sac type organisation in pollen grain. This is called Nemec phenomenon.
(iii) Smallest pollen grain: Myosotis (2.5 -3.5 µ).
(iv) Largest pollen grain : Mirabilis (250 µ).
(v) Longest or Filamentous pollen grain : Zostera (2500 µ).
(e) Development of Male Gametophyte
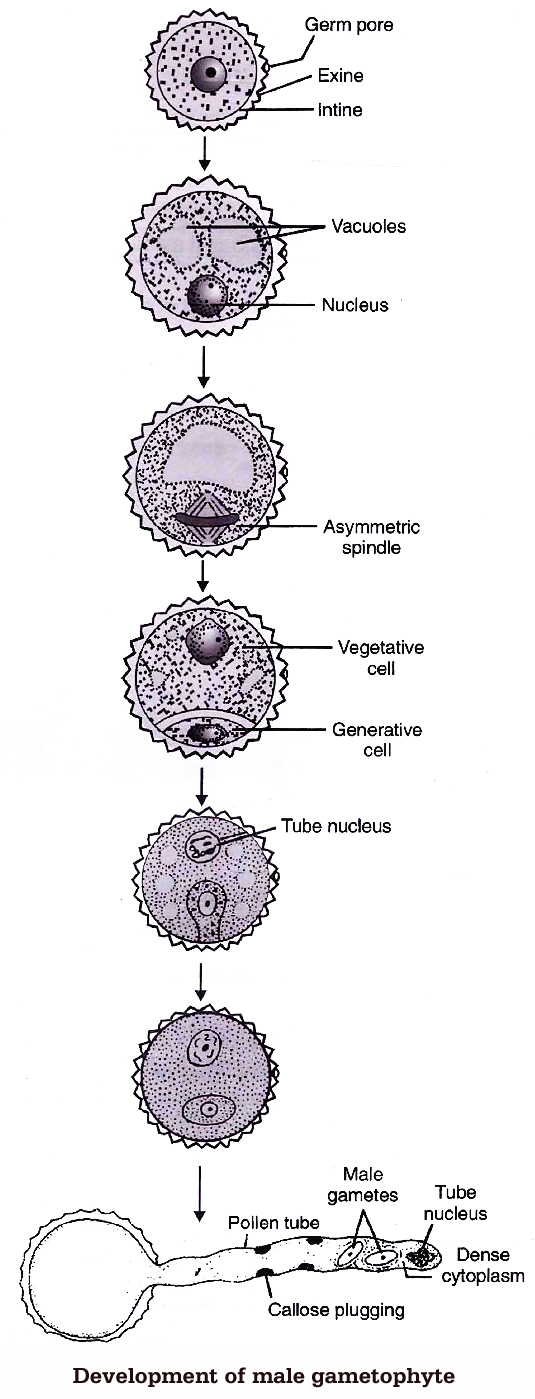
The microspore is the first cell of male gametophyte. The germination of microspore starts in situ (in the mother place). Microspore may be best defined as partially developed male gametophyte.
Microspores divide mitotically into large tube cell and small generative cell Pollination takes place at two celled stage in 60% of angiosperms (in some cases at three-celled stage). The further development of these male gametophyte takes place on the stigma.
Pollen grain expands by absorbing the liquid from the moist surface of stigma. Stigma provides, boron, sugar, amino acids etc. The intine comes out in the form of pollen tube, from germ pores.
The pollen grains are either monosiphonous (with one pollen tube) or polysiphonous (with more than one pollen tubes), e.g., members of Cucurbitaceae and Malvaceae.
The generative nucleus divides mitotically to form two male gametes called sperm. The male gametes are non-motile and amoeboid. They are slightly unequal in size, such a pollen will be called three celled pollen or mature male gametophyte.
The function of pollen tube is to carry aflagellated sperm. Tube nucleus enters first in the pollen tube and is a vestigial structure and soon disintegrates. Growth of the pollen tube is chemotropic apicle and entire cytoplasm of pollen grain is confined to tip of the pollen tube.
Concept Builder
(i) The pollen tube was first observed by G.B. Amici (1824) in Portulaca.
(ii) Longest pollen tube occurs in Zea mays.
(iii) B-Ca-inositol sugar complex act as chemotropic agent for pollen tube growth.
The Pistil, Ovule and Embryo sac
(a) Pistil (Female Sex Organ)
The gynoecium may consist of a single pistil (monocarpellary) or may have more than one pistil (multicarpellary).
If there are more than one, the Pistils may be fused together (Syncarpous) or may be free (apocarpous).
There are three parts of each pistil -the stigma, style and ovary. The stigma is upper broader region which is specialised for receiving pollen grains.
The style is long stalk-like structure and ovary is basal swollen ovule containing region.
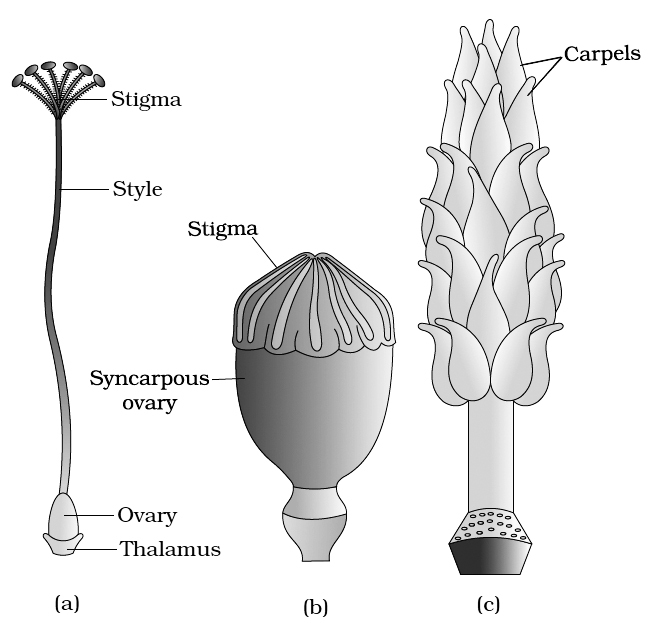
(c) A multicarpellary, apocarpous gynoecium of Michelia
(b) Ovule (Integumented Megasporangium)
(i) Ovary has an ovarian cavity with one or more chambers (lobules). The placenta is located inside the ovarian cavity. One or many ovules are present inside the ovary.
Plants with one ovule in an ovary: (i) Wheat, (ii) Paddy, (iii) Mango
Plants with many ovules in an ovary: (i) Papaya, (ii) Water melon, (iii) Orchids
(ii) The ovule is a small structure attached to the placenta by means of a stalk called funicle. The point of attachment of the funicle with the main body of the ovule is called hilum. Thus, hilum represents the junction between ovule and funicle. Sometimes funicle gets fused with the body of the ovule along one side and forms a ridge known as raphe. The basal region of the ovule is known as Chalaza.
(iii) Main body of an ovule is called nucellus (megasporangium) which consists of a mass of parenchymatous tissue. Cells of nucellus have abundant reserve food materials. Depending upon the development of nucellus, ovules are of two types:

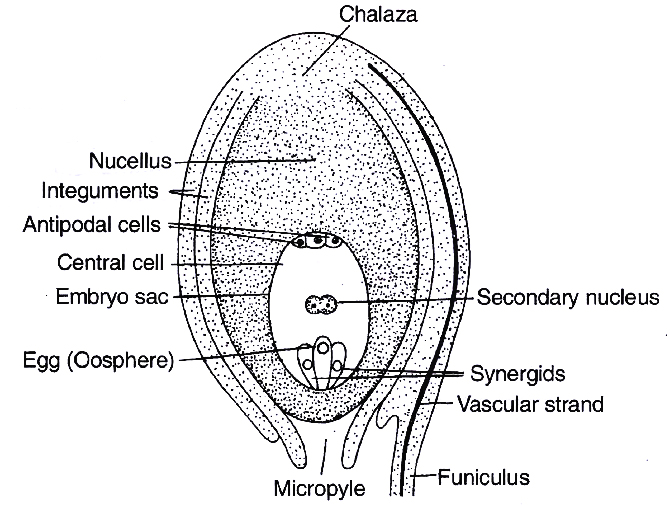
containing developed embryo sac (female gametophyte)
(iv) The nucellus is invested all around by one or two layered protective covering called integuments, except apex where a small passage is formed known as micropyle. On the basis of number of integuments, ovules are of following types:
(a) Unitegmic: Ovules with one integument, e.g., members of gamopetalae and gymnosperms.
(b) Bitegmic : Ovules with two integuments, e.g., members of polypetalae and monocots.
(c) Ategmic : Ovules are without integument, e.g., Santalum, Loranthus (Parasites) and Liriosoma.
(v) On the basis of the relative position of micropyle, body of the ovule and funicle, i.e., the degree of curvature, there are six types of ovules:
(a) Orthotropous : The micropyle, chalaza and funicle are in a straight line. This is the most primitive type of ovule, e.g., Piper, Polygonum, Cycas.
(b) Anatropous : The ovule turns 180º angle. Thus it is inverted ovule. Micropyle lies close to hilum or at side of hilum, e.g., found in 82% of angiosperm families.
(c) Hemianatropous: Ovule turns at 90º angle upon the funicle or body of ovule is at right angle to the funicle, e.g., Ranunculus.
(d) Campylotropous: Ovule is curved more or less at right angle to funicle. Micropylar end is bend down slightly, e.g., in members of Leguminosae, Cruciferae.
(e) Amphitropous: Ovule as well as embryo sac is curved like horse shoe, e.g., Lemna, Poppy, Alisma.
(f) Circinotropous: The ovule turns at more than 360º angle, so funicle becomes coiled around the ovule, e.g., Opuntia (Cactaceae), Plumbaginaceae.
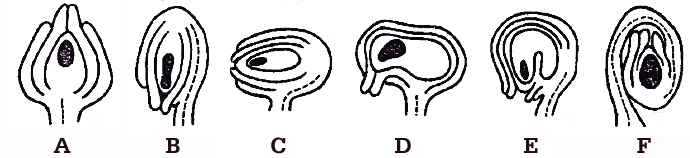
A. Orthotropous, B. Anatropous, C. Hemianatropous,
D. Campylotropous, E. Amphitropous, F. Circinotropous
Concept Builder
(i) Third integument in the form of aril develops from base of ovule or funicle in many plants, e.g., Litchi, Asphodelus and Inga dulce. In litchi and Inga dulce, aril is fleshy and edible.
(ii) In some ovules, e.g., Zostera, a group of thickened cells is found in the chalazal region just below the embryo sac, called hypostase. When a similar group of specialized cells is found in the nucellar region just above the embryo sac, it is called epistase.
(iii) In castor (Ricinus), proliferation of the outer integumentary cells at micropylar region is called caruncle or strophiole. It performs two functions :
a. It acts as water absorbing pad.
b. It is made up of sugary substance that attract and helps in the seed dispersal by ants (myrmecochory).
(iv) The placental or funicular outgrowth present at, the micropylar end is called obturator. It directs the passage of pollen tube towards the ovule.
(c) Megasporogenesis
(i) Any of the cells of nucellus hypodermis towards the micropylar end gets differentiated from the other cells. This cell is called archesporial cell. In crassinucellate ovules, it undergoes periclinal division to form an outer primary parietal cell and inner primary sporogenous cell. The later behaves as megaspore mother cell (MMC). The archesporial cell directly behaves as megaspore mother cell in tenuinucellate ovules.
(ii) The megaspore mother cell is large sized containing dense cytoplasm and a prominent nucleus. Some carbohydrate storing bodies appear between cell wall and plasma membrane of megaspore mother cell in Lilium, called paramular bodies.
(iii) The MMC (2n) undergoes meiosis and forms a linear tetrad of 4 haploid megaspores. The process of formation of megaspores from the MMC is called megasporogenesis.
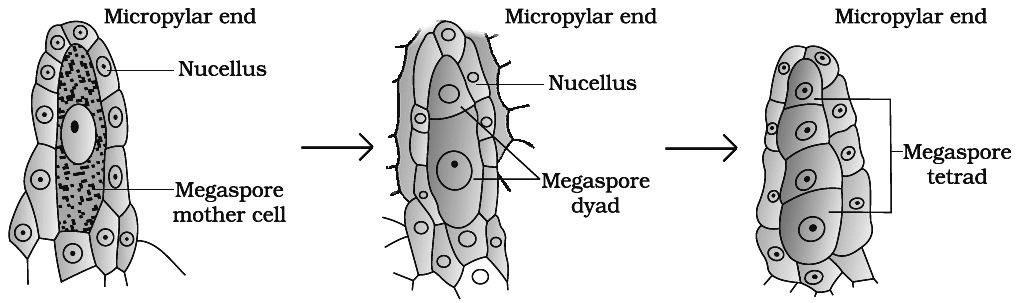
(d) Female Gametophyte or Embryo sac
(i) P. Maheshwari classified the embryo sac on the basis of number of megaspore nuclei participating in embryo sac formation into following types :
Monosporic embryo sac : Only one megaspore nucleus forms embryo sac, e.g., Polygonum, Oenothera.
Bisporic embryo sac: Two megaspore nuclei take part in development of embryo sac, e.g., Allium, Endymion.
Tetrasporic embryo sac: All the four megaspore nuclei take part in development of embryo sac, e.g., Adoxa, Plumbago, Drusa, Fritillaria, Paenaea, Plumbagella, Peperomia.
(ii) Development of Monosporic Embryo sac (Polygonum type)
(A) In majority of angiosperms, one of the megaspore is functional while the other three degenrate. Only the functional megaspore (n) develops into the female gametophyte. This process of embryo sac formation from a single megaspore is termed monosporic development.
(B) Polygonum type of embryo sac is found in 80% flowering plants. This development has been studied in (Polygonum by Strasburger). The nucleus of chalazal functional megaspore (4th from micropyle) divides by three mitotic divisions to form 8 nuclei, four towards each pole. One nucleus from each pole moves to the middle and they form polar nuclei. These mitotic divisions are strictly free nuclear, i.e., nuclear divisions are not followed immediately by cell wall formation.
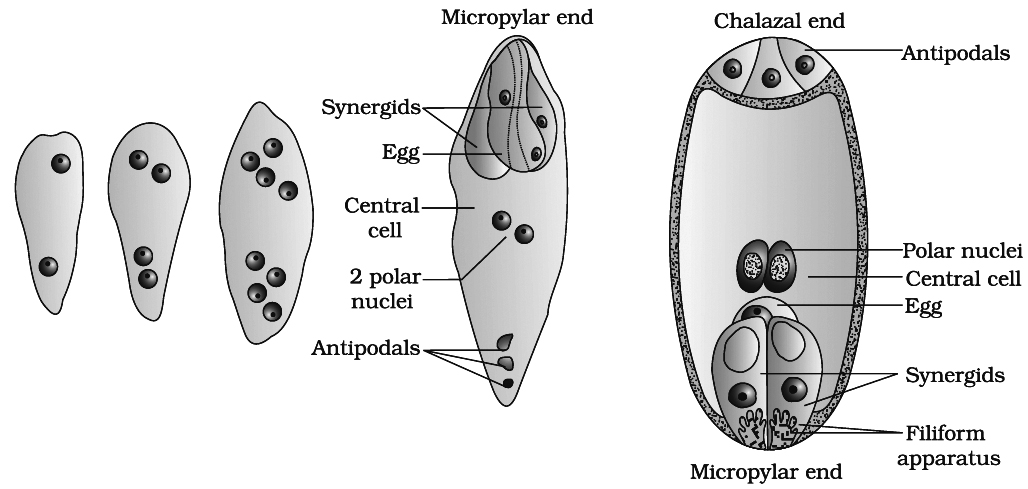
At this stage, following changes occur:
(a) Three of the nuclei (n) get organised as cells at micropylar end forming egg apparatus. One is the egg cell (n) and two are synergids (n).
(b) Three nuclei get organised as antipodal cells (n) at chalazal end
(c) Two nuclei in the centre are called polar nuclei (n)
This constitutes a 7-celled and 8-nucleated embryo sac.
Organization of Embryo Sac
(a) Synergids or helper cells or co-operative cells: These cells generally possess a micropylar nucleus and a chalazal vacuole. The electron microscopic studies have revealed that the synergids lack a cell wall on their chalazal side at maturity. They are characterised by the presence of a 'filiform apparatus at the' micropylar tip. It is in the form of finger like projections, each projection comprising a core of microfibrils enclosed in a sheath. Usually one synergid starts to degenerate just with pollination. The synergids perhaps secrete some chemotropic substance and thus, direct the pollen tube growth inside embryo sac.
(b) Egg : The egg shows cytoplasmic polarity opposite to synergid and its wall is thicker at the micropylar end. Usually the egg has a micropylar vacuole and a chalazal nucleus. Plasmodesmata connection is present in between egg and synergids.
(c) Antipodals or vegetative cells : These are vegetative cells of embryo sac. In most of the plants there are three antipodal cells.
(d) Central Cell: It is the largest cell of the embryo sac. It initially contains two polar nuclei which fuse just before, fertilization to form a secondary nucleus or definitive nucleus (2n).
Concept Builder
(i) Synergids are absent in Plumbago and Plumbagella. Number of synergid is one in Peperomia.
(ii) In Zea mays 20 antipodal cells are present, in Sasa paniculata they are 300 in number. This is perhaps the highest number recorded for any plant. These are absent in Oenothera type.
pollination
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
POLLINATION
In flowering plants, male and female gametes are produced in the pollen grain and embryo sac respectively. These gametes are non-motile, therefore, they have to be brought together for fertilisation. Pollination is the mechanism to achieve this objective. The transfer of pollen grains to the stigma is called pollination. Depending upon the source of pollen, pollination is of three types :
(1) Autogamy :
If the pollen grains are transferred from anther to the stigma of the same flower, the process is called self pollination or autogamy.
Contrivances for autogamy
(i) Bisexuality - Flower should be bisexual, e.g., Catharanthus
(ii) Homogamy - Male and female reproductive parts in a bisexual flower mature at the same time, e.g., Mirabilis.
(iii) Cleistogamy - Sometimes bisexual flowers remain closed and never open, such flowers are known as cleistogamous. In such flowers, the anthers and stigma lie close to each other. When the anthers dehisce, the pollen grains come in contact with stigma and pollination takes place. Thus, cleistogamous flowers are invariably autogamous as there is no chance of cross-pollen landing on the stigma. So one of the advantage of cleistogamy is, it ensures seed-set even in the absence of pollinators, e.g., Arachis hypogea (Groundnut). Some plants like Viola (common pansy), Oxalis and Commelina produce chasmogamous flowers (open flowers) as well as cleistogamous flowers.
(iv) Bud pollination -When self pollination occurs in the bud stage before the opening of flowers, e.g., Pisum, Wheat, Rice.
(2) Geitonogamy:
Pollination taking place between the two flowers of the same plant (genetically self pollination but ecologically or functionally cross pollination), e.g., Maize.
(3) Xenogamy :
When the pollen grains are transferred from the anther to the stigma of the flower of a different plant, it is called as xenogamy. (It is cross pollination, both genetically and ecologically).
Outbreeding Devices (Contrivances for cross pollination)
Majority of flowering plants produce hermaphrodite flowers and pollen grains are likely to come in contact with the stigma of the same flower. Continued self-pollination results in inbreeding depression, therefore angiosperms have developed following devices to, discourage self pollination and to encourage cross-pollination.
(i) Dichogamy: In bisexual flowers, the two sexes mature at different timings. When anthers mature first it is called protandry, e.g., Sunflower, Cotton, Salvia. When gynoecium matures first it is called as protogyny, e.g., Ficus, Aristolochia, Magnolia.
(ii) Incompatibility: It is the inability of certain gametes, even from genetically similar plant species, to fuse with each other. This is also called intra-specific incompatibility, self-sterility or self-incompatibility.
|
Concept Builder
|

Incompatibility may involve morphological or physiological mechanisms, therefore there are two types of this device :
(a) Morphological self-incompatibility : It occurs in flowers having heterostyly. Flowers are dimorphic or trimorphic with regard to the length of style. Thus facilitate cross pollination.
e.g., Primula (Primrose), Jasminum, Lythrum

(b) Physiological self-incompatibility :
It is controlled by multiple alleles of S-gene. There are two types of physiological self-incompatibility :
(i) If incompatibility is due to the genotype of the sporophytic/stigmatic tissues, is termed sporophytic incompatibility (SSI), e.g., Brassicaceae, Asteraceae. (ii) The incompatibility due to the genotype of the pollen is termed gametophytic incompatibility (GSI), e.g., Solanaceae, Liliaceae, Poaceae. This may be due to prevention of pollen germination, deorientation of pollen tube, or even failure of nuclear fusion. A plant carries two such alleles e.g., S1S2, S2S3, S1S3, S2S4, S3S5. A pollen carries only one allele. If it happens to be one of the two alleles of pistil, the rejection may occur on stigma surface (SSI) or in the style (GSI).
(iii) Unisexuality or dicliny : It is the formation of unisexual flowers. Thus, cross pollination becomes obligatory. It can be seen in monoecious plants where both male and female flowers are produced on same plant, e.g., maize, castor as well as dioecious plants where male and female flowers are produced on different plants, e.g., Vallisneria, Papaya. Monoecious condition prevents autogamy but not geitonogamy while dioecious condition prevents both autogamy and geitonogamy.
(iv) Herkogamy : It is the presence of natural and physical barrier between androecium and gynoecium, e.g., in Calotropis, gynoecium is fused with pollinium (anthers) and form gynostegium.
(v) Prepotency : Pollen grain of one flower germinates more rapidly over stigma of another flower as compared to its own flower, e.g., Apple, Grape.
Agencies for cross pollination :
Pollinating agency may be biotic (animals) or abiotic (wind and water). Majority of plants use biotic agents for pollination.
A. Abiotic agents
(a) Anemophily: (Pollination by wind).
 It is common amongst abiotic pollinations. It is a non-directional and wasteful process. The female flowers have large, feathery or brush like stigmas to catch the pollen grains. Anemophilous flowers are small and inconspicuous with long and versatile stamens. Pollen grain are dry, powdery, light and non-sticky. The flowers often have a single ovule in each ovary and numerous flowers packed into an inflorescence. Anemophily is quite common in grasses.
It is common amongst abiotic pollinations. It is a non-directional and wasteful process. The female flowers have large, feathery or brush like stigmas to catch the pollen grains. Anemophilous flowers are small and inconspicuous with long and versatile stamens. Pollen grain are dry, powdery, light and non-sticky. The flowers often have a single ovule in each ovary and numerous flowers packed into an inflorescence. Anemophily is quite common in grasses.
e.g., Maize, wheat, sugarcane, bamboo, Pinus (winged pollen), Papaya.
(b) Hydrophily : (Pollination by water).
It is quite rare in angiosperms and is limited to about 30 genera, mostly monocotyledons. All aquatic plants are not hydrophilous. In a majority of aquatic plants like water hyacinth and water lily, the flowers emerge above the level of water and are pollinated by insects or wind.
|
Hydrophyte |
Type of pollination |
|
1. Alisma |
Entomophily |
|
2. Lotus |
Entomophily |
|
3. Potamogeton |
Anemophily |
|
4. Myriophyllum |
Anemophily |
Hydrophily is of two types:
(1) Epihydrophily : Pollination at the surface of water
Example : Vallisneria (Tape grass, Ribbon weed)
Vallisneria is a dioecious rooted submerged aquatic plant in which male flowers are small and light weight. Female flowers have very long coiled pedicels which uncoil when they become mature. Male flowers float at the surface of water. As soon as the male flowers touch the female flowers, anther lobes burst, stigma receives the pollen grains and pedicels coil again.

(2) Hypohydrophily : Pollination inside the water.
Examples: Zostera (sea grass), Ceratophyllum
Zostera is a marine water plant. Female flowers remain submerged in water and the pollen grains are long, ribbon like and they are carried passively inside the water, some of them reach the stigma and achieve pollination.
(i) Pollen grains are protected from wetting by a mucilaginous covering in most of the hydrophilous species.
(ii) Anemophilous as well as hydrophilous flowers are not very colourful and do not produce nectar.
(iii) Pollen grains coming in contact with the stigma is a chance factor in both wind and water pollinating plants. To compensate for these uncertainities and associated loss of pollen grains, the flowers produce enormous amount of pollen when compared to the number of ovules available for pollination.
B. Biotic Agents
(a) Entomophily: (Pollination by insects)
(1) 80% pollination occurs by insects (chief pollinators) and honey bee is main pollinator among insects. All the flowers pollinated by bees are brightly coloured, have a sweet smell and produce nectar. Entomophilous flowers produce a small amount of pollen which have a spinous and may have a sticky exine due to presence of pollenkitt. The stigmas of such flowers are long, rough and sticky. If the entomophilous flowers are small, a number of flowers are clustered into an inflorescence to make them conspicuous. Moth pollinated plants are white flowered and fragrant. The flowers pollinated by flies and beetles secrete foul odours to attract these animals.
(2) When petals are not conspicuous, other parts may become coloured or showy to attract the insects, e.g., bracts in Bougainvillea, leaves in Euphorbia pulcherrima (Poinsettia), one sepal in Mussaenda acts like advertisement flag, anthers are coloured in Mimosa.
(3) To sustain insect visits, the flowers have to provide rewards to the insects. Nectar and pollen grains are the usual floral rewards. In plants like Papaver, Rosa, Clematis, etc., edible pollen grains are produced. Some of the pollen grains stick to the back of insects while feeding on the edible pollen grains. Some species provide safe place to insect for laying eggs, e.g., Amorphophallus (the flower is 6 feet in height), Yucca.
(4) Some special cases of entomophily are as follows:
(i) Yucca : The flowers at Yucca and pollinating moth, i.e., Pronuba yuccasella / Tegaticula show a very close relationship. The insect punctures the ovary for egg laying. The insect creeps on the style and deposits pollen ball in between the stigmatic lobes, hence bringing about pollination. The larvae of the moth come out of the eggs as the seeds start developing.
(ii) Salvia or Sage plant : It shows lever mechanism or turn pipe mechanism. Anthers are distractile; lower lobe is sterile and upper lobe is fertile. The flower is bilabiate and protandrous. The insect sits on the lower lobe of the corolla; the upper fertile lobe of anther touches the body of insect. It ruptures and pollen grains are shed on the back of the insect. When this insect visits the other flowers, pollination is affected.

(iii) Ficus sp. : Trap Door mechanism in Ficus carica. The receptacle forms a cup having a cavity. The cavity has a pore called ostlole near which male flowers are present. Female flowers are present at the bottom and gall (sterile) flowers are present between these types. The pollinating insect is Blastophaga or gall insect. The insect lays eggs in the bottom, the larvae feed on the ovules of gall flowers. When the young insects crawl out of the inflorescence, their bodies are laden with pollen grains. They enter new hypanthodium and affects the pollination.

(iv) Calotropis : It shows translator or clip mechanism. Pollen grains are present in pollinia. The pollinia are attached to a rough and sticky disc called corpusculum. When the insect visits the flower, pollinia get entangled in the legs of the insect. When this insect visits other flowers, pollinia are transferred.

(v) Centaurea : It belongs to Compositae family and exhibits piston mechanism. It has syngenesious and epipetalous androecium. The anther lobes dehisce introsely. When filaments contract on being touched by the insect, the stigma pushes the pollen grains out of the anther tube which stick to the body of the insect. When the insect visits some other flower, pollination is affected.
(vi) Aristolochia : The upwardly directed protogynous flowers have downwardly directed stiff hairs in the middle of the corolla tube. These form the trap. This is called pit fall mechanism or fly trap mechanism.

(vii) Orchid : Pollination occurs by wasp (Pseudocopulation mechanism), e.g. , Ophrys (orchid) and Colpa (wasp). The flower of Ophrys resemble in shape, colour and odour to female of wasp Colpa aurea, thus showing mimicry. It is a case of co-evolution also.
(5) Many insects may consume pollens or the nectar without bringing about pollination. Such floral visitors are referred to as pollen/nectar robbers.
Pollen robbers create a hole at the base of corolla tube and draw nectar from a flower whose design is not suitable for them. Other insects often take advantage of the same.
(b) Ornithophily: (Pollination by birds).
Flowers are brightly coloured but odourless and produce plenty of nectar and large quantity of pollens, e.g., Bombax (red silk cotton), Callistemon (bottle brush), Sterlitzia, Erythrina (coral tree) etc. Honey bird, humming bird and sun bird are common pollinators.
(c) Cheiropterophily: (Pollination by bats).
Bats pollinate the flowers of tropical regions, e.g., Anthocephallus (Kadamb), Kigelia (Sausage tree), Adansonia (Baobab tree).
(d) Malacophily: (Pollination by snails), e.g., Arum; Lilies, Arisaema, Lemna.
(e) Ophiophily: (Pollination by snakes), e.g., Santalum, Michelia.
(f) Some larger animals have been reported as pollinators in some species, e.g., Lemur (Primate) in Ravenela plant, Lizard in Flax
|
Self Assessment
|

Q.21 Polygonum type of embryo sac is
(1) 8-celled (2) 15-nucleated (3) Haploid (4) Exosporic type
Q.22 Mark the odd option (w.r.t. contrivances of autogamy)
(1) Homogamy (2) Cleistogamy (3) Dicliny (4) Bud pollination
Q.23 Which of the following pollination is common amongst abiotic agents?
(1) Hydrophily (2) Entomophily (3) Ornithophily (4) Anemophily
Q.24 Epihydrophily is found in
(1) Tape grass (2) Sea grass (3) Lotus (4) Alisma
Q.25 Mark the incorrect match (w.r.t. pollination)
(1) Salvia Turn pipe mechanism (2) Ficus Fly trap mechanism
(3) Aristolochia Pit fall mechanism (4) Calotropis Clip mechanism
Q.26 Which of the following plant provide safe place to insect for laying eggs?
(1) Sage plant (2) Ophrys (3) Centaurea (4) Amorphophallus
Q.27 Pollination occurs by pseudocopulation mechanism in
(1) Ophrys (2) Fig (3) Mango (4) Zea mays
Q.28 Lemur, a large animal, acts as pollinator in
(1) Flax (2) Ravenela (3) Capsella (4) Hydrilla
Q.29 Which of the following type of pollination is present in Santalum?
(1) Ornithophily (2) Ophiophily (3) Malacophily (4) Entomophily
Q.30 In ornithophily, flowers are
(1) Dull coloured (2) Nectarless (3) Odourless (4) Very small
Ans. Q.21 (3), Q.22 (3), Q.23 (4), Q.24 (1), Q.25 (2), Q.26 (4), Q.27 (1), Q.28 (2), Q.29 (2),
Q.30 (3)
POLLEN-PISTIL INTERACTION
Pollination does not guarantee the transfer of the right type of pollen. Often, pollen of the wrong type, either from other species or from the same plant (if it is self-incompatible), also land on the stigma.
The pistil has the ability to recognise the pollen, whether it is of the right type (compatible) or the wrong type (incompatible). The ability of the pistil to recognise the pollen followed by its acceptance or rejection is the result of a continuous dialogue between pollen and the pistil.
This dialogue is mediated by chemical components of the pollen interacting with those of the pistil. If the reaction is favourable, the pollen grain germinates on the stigma to produce pollen-tube through one of the germ pores.
Plants in which the pollen grain are shed at 2-celled stage, the generative cell divides and forms the 2 male gametes during the growth of pollen tube in the stigma. If pollen grains are shed at 3-celled stage, pollen tubes carry the 2-male gametes from the beginning.
(1) Entry of the pollen tube into an ovule
(A) Pollen tube, after reaching the ovary, enters the ovule through the micropyle. It is called as porogamy as seen in most of the flowering plants.
(B) In some plants, e.g., Casuarina, the pollen tube enters an ovule through chalaza and it is called as chalazogamy.

(a) Pollen grains germinating on the stigma; (b) Pollen tubes growing through the
style; (c) L.S. of pistil showing path of pollen tube growth
(C) Some times the pollen tube enters into an ovule through integuments or funicle. It is called as mesogamy. e.g,. Cucurbita.
(2) Entry of the pollen tube into the embryo sac :
Irrespective of the place of entry of pollen tube into the ovule, the tube invariably enters the embryo sac at micropylar end, i.e., degenerating synergid cell. Entry of pollen tube in the embryo sac is under chemotropic guidance (synergids have filiform apparatus and secrete some chemicals).

(A) enlarged view of an egg apparatus showing entry of pollen tube into a synergid;
(B) Discharge of malegametes into a synergid and the movements of the sperms,
one into the egg and the other into the central cell
(3) All these events from pollen deposition on the stigma until pollen tubes enter the ovule are together referred to as pollen-pistil interaction. This interaction is a dynamic process.
(4) Pollen germination can be studied by dusting pollen (e.g., pea, chick pea, Grotalaria, balsam, Vinca) on a glass slide containing a drop of 10% sugar solution with boric acid, Ca, Mg and K salts. After 15-30 minutes, pollen tubes will' be observed to come out of the pollen grains. So, this germination of pollen grain in laboratory is called hanging drop method.
(5) Artificial hybridisation : It is one of the major approaches of crop improvement in which crosses are made between different varieties, species and genera in order to combine their desirable characters in a single superior variety. Emasculation and bagging are two precautionary measures in this hybridisation. Emasculation is the removal of anthers from the floral buds of female parent bearing bisexual flowers. Bagging is the covering of both emasculated as well as non-emasculated flowers with butter paper or polythene to prevent contamination of its stigma with unwanted pollen. When the stigma of bagged flower attains receptivity, mature pollen grains collected from anthers of the male parent are dusted on the stigma and the flowers are rebagged, and the fruits allowed to develop.
double fertilization
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
DOUBLE FERTILIZATION
Pollen tube enters the degenerating synergid and bursts, thereby releasing two male gametes. One
male gamete fuses with the egg to form diploid zygote. This fusion is called syngamy or
generative fertilization.
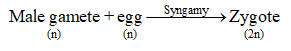
The other male gamete fuses with secondary nucleus (2n, i.e., fused polar nuclei of both polar
nuclei) to form primary endosperm nucleus (3n). This fusion is also called triple fusion because
three nuclei takes part in this fusion. It is also known as vegetative fertilization or
pseudofertilization or trophomixis.
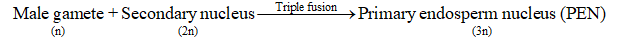
Since two types of fusions, i.e., syngamy and triple fusion take place in an embryo sac the
phenomenon is termed double fertilisation. This event occurs in flowering plants only. Five nuclei
are involved in double fertilization.
Concept Builder
(i) The syngamy was discovered by Strasburger.
(ii) Triple fusion and double fertilization was discovered by S.G. Nawaschin and Guignard in Lilium and Fritillaria.
(iii) Entry of more than one pollen tubes in an ovule leading to the occurrence of supernumerary male nuclei (polyspermy).
(iv) When two pollen tubes enter an ovule and release their contents, it is possible that the egg may be fertilized by male gamete from one tube and triple fusion may involve participation of male gamete from another tube. This phenomenon is called as heterofertilization, e.g., Zea mays.
(v) When the entry of male gamete is not accompanied by fusion,the phenomenon is called as semigamy.
(vi) X-bodies are darkly stained DNA containing bodies found in synergid receiving pollen tube.
(vii) The percentage of pollen germination and tube growth is better in larger populations. This is referred to as "population effect" or "crowding effect".
post fertilization
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
POST-FERTILIZATION: Structures and Events
Development of endosperm and embryo, maturation of ovules into seeds and ovary into fruit are collectively termed as post-fertilization events.
A. Endosperm
This is a product of triple fusion and develops from central cell of embryo sac. It is generally a triploid tissue. The cells of this tissue are filled with reserve food materials and are used for the nutrition of the developing embryo.
It is absent in families such as Orchidaceae, Podostemaceae and Trapaceae.
The stony endosperm is present in betel nut (Areca nut) and date palm (Phoenix dactylifera).
Diploid endosperm is found in Oenothera.
Types of endosperm :
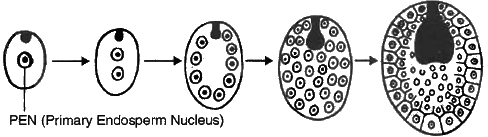
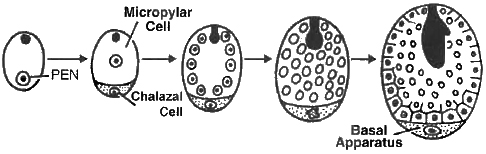
(i) Nuclear Endosperm : It is the most common type of endosperm in angiospermic plants. During the development of this endosperm, at first PEN undergoes successive nuclear divisions without wall formation (free nuclear division). In this manner, large number of free nuclei are produced. Finally, wall formation begins and it makes the endosperm a multicellular tissue, e.g., Cotton, Maize, Capsella, Coconut (milk).
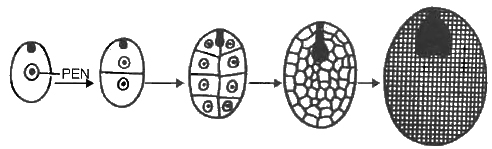
(ii) Cellular Endosperm: Primary endosperm nucleus divides many times and each division is followed by wall formation, e.g., Petunia, Utricularia, Coconut (copra).
(iii) Helobial Endosperm: It is an intermediate between nuclear and cellular type, e.g., members of order helobiales (Monocot).
Concept Builder
(i) The seeds with double endosperm is found in coconut (Cocos nucifera), (a) liquid endosperm, (b) cellular endosperm.
(ii) Xenia : The effect of foreign pollen on endosperm character is called xenia. This term was given by Focke. This was first observed in maize endosperm colour.
(iii) Metaxenia : Discovered by Swingle. The effect of foreign pollen on somatic tissue lying outside the endosperm is known as metaxenia, e.g., in date palm size of fruits and maturity time depends upon foreign pollen. .
(iv) Ruminate Endosperm : Endosperm with irregular surface, also known as chewed endosperm, e.g., Passiflora, Annona, Myristica.
(v) Mosaic Endosperm : Endosperm with sugary and starchy parts forming different colour patches of yellow and white, e.g., Maize.
B. Embryo
The development of an embryo from a zygote is called embryogeny. Embryo develops at the micropylar end of the embryo sac where the zygote is situated. Most zygotes divide only after certain amount of endosperm is formed.
This is an adaptation to provide assured nutrition to the developing embryo. It means endosperm development precedes embryo development.
Early stages of embryogeny are similar in both monocotyledons and dicotyledons.
(a) Development of Embryo in Dicots
The normal type of dicot embryo development has been studied in Shepherd's purse (Capsella bursa-pastoris) which belongs to family Cruciferae.
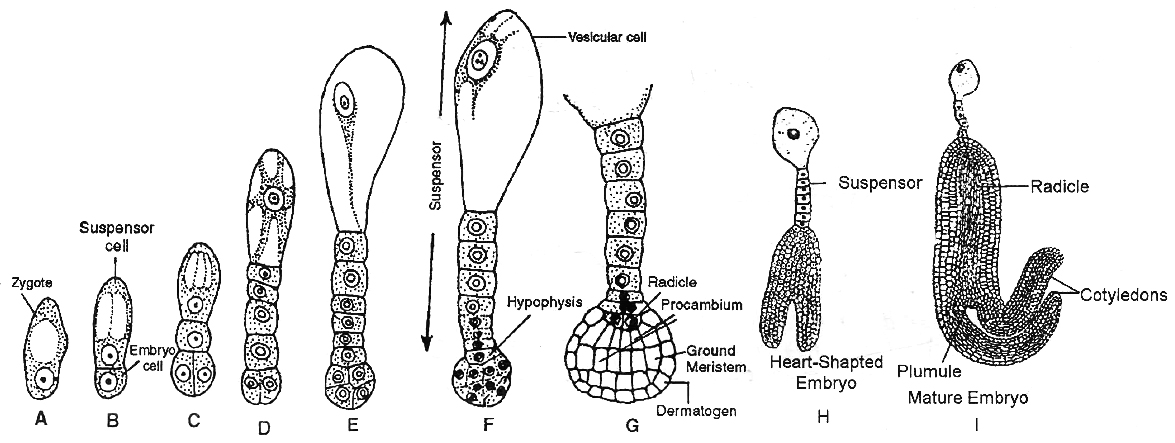
This is called as Crucifer or Onagrad type of embryo development. The development of embryo is endoscopic. Zygote (oospore) divides into two unequal cells, larger suspensor cell towards micropyle and a smaller embryonal cell (terminal cell) towards antipodal region.
The suspensor cell undergoes transverse divisions forming 6-10 celled long suspensor. The first cell of the suspensor (towards micropyle) is large and called haustorium or vesicular cell.
The last cell of suspensor (towards embryo cell) is known as hypophysis. It forms radicle tip.
Embryonal cell divides twice vertically and once transversely to produce a two-tiered eight-celled embryo. The epibasal tier forms two cotyledons and a plumule while the hypobasal tier produces only hypocotyl and most of the radicle.
For this the octant embryo undergoes periclinal divisions producing protoderm, procambium and ground meristem. It is initially globular but with the growth of cotyledons it becomes heart-shaped and then assumes the typical shape, e.g., Capsella bursa-pastoris.
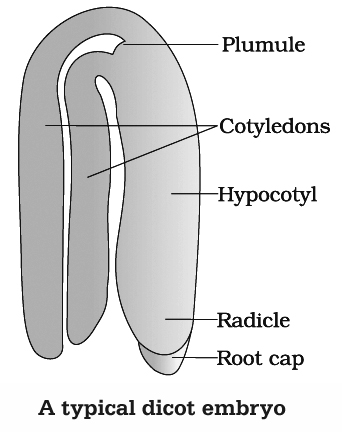
A typical dicot embryo consists of an embryonal axis and two cotyledons. The portion of embryonal axis above the level of cotyledons is the epicotyl, which terminates with the plumule (stem tip).
The cylindrical portion below the level of cotyledons is hypocotyl that terminates, at its lower end in the radicle. In orchids, Orobanche and Utricularia, the embryo does not show distinction of plumule, cotyledons and radicle.
(b) Development of Embryo in Monocot
The normal type of monocot embryo development has been studied in Luzula forsteri and is called Segittaria type. Suspensor is single celled in monocots.
The zygote of oospore divides transversely producing a vesicular suspensor cell towards micropylar end and embryo cell towards the chalazal end.
The embryo cell divides transversely again into a terminal and a middle cell. The terminal cell divides vertically and transversely into globular embryo.
It forms a massive cotyledon and a plumule.
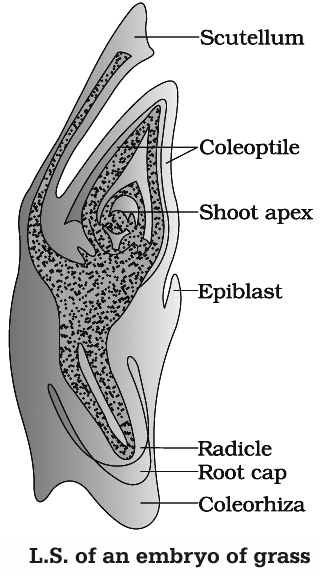
Growth of cotyledon pushes the plumule to one side. Remains of second cotyledon occur in some grasses. It is called epiblast. The single cotyledon of monocots is called scutellum. It is shield shaped and lateral in position but appears terminal.
The middle cell gives rise to hypocotyl and radicle. Radicle is enclosed in an undifferentiated sheath called coleorhiza. Epicotyl has a shoot apex and a few leaf primordia enclosed in a hollow foliar structure, the coleoptile.
C. Seed
It is often described as fertilised ovule. A seed typically consists of seed coat(s), cotyledon(s) and an embryonal axis. Mature seeds are of two types:
(a) Non endospermic or exalbuminous seeds:
In seeds like gram, pea, groundnut etc. the endosperm is completely consumed by developing embryo, thus the seeds are called non-endospermic or exalbuminous.
(b) Endospermic or albuminous seed :
In monocots and castor bean (dicots) embryo does not consume all endosperm. So, it persists in the mature seed. Such seeds are called endospermic or albuminous seeds. In these seeds, food is stored in endoseprm, e.g., wheat, maize, barley, sunflower, coconut.
Mostly nucellus is consumed after fertilisation due to absorption of food by the endosperm and embryo, but in some seeds remnants of nucellus are of persistant nature.
This residual, persistent nucellus is called perisperm. Such seeds are called perispermic seeds, e.g., Black pepper (Piper nigrum), beet.
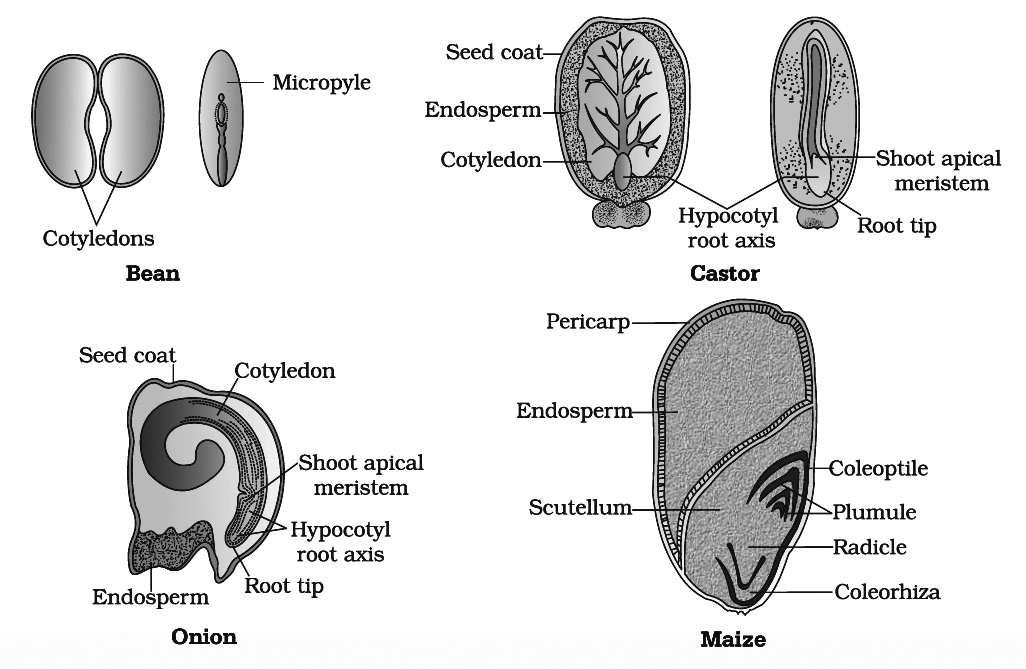
Integuments of ovules harden as tough protective seed coats. The outer seed coat called testa develops from outer integument and inner seed coat called tegmen is derived from inner integument.
The micropyle remains as a small pore in the seed coat. Micropyle facilitates entry of oxygen and water into the seed during germination.
As the seed matures, its water content is reduced and seeds become relatively dry (10-15% moisture by mass).
The embryo may enter a state of inactivity called dormancy or if favourable conditions like adequate moisture, oxygen and suitable temperature are available, they germinate.
The transformation of ovules into seeds and ovary into fruit proceeds simultaneously. The ovary wall develops into the fruit wall, called pericarp.
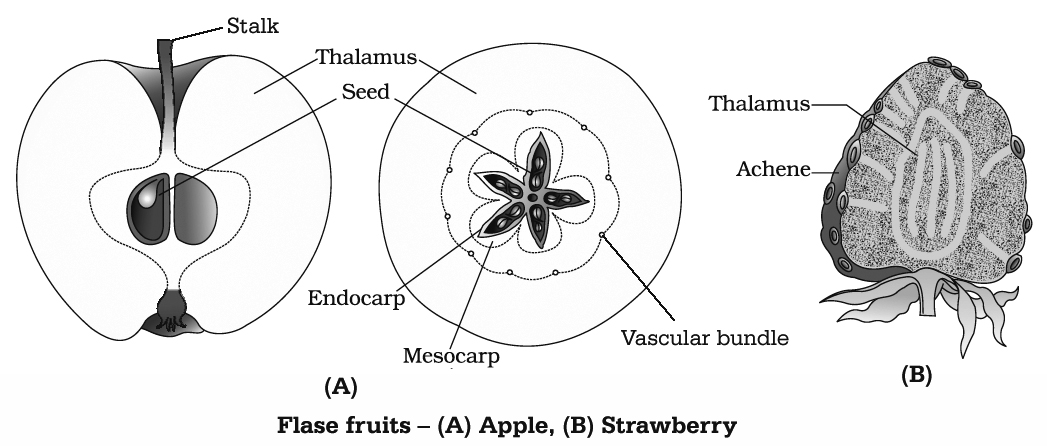
The fruit may be fleshy (guava, orange, mango) or dry (groundnut, mustard). In some cases thalamus and other floral parts show proliferation alongwith development of ovary wall.
Such fruits are called false fruits, e.g., Apple, Strawberry, Cashew nut. Most fruits develop from the ovary and are called true fruits.
In some plant species, fruits develop without fertilisation. Such fruits are seedless and are called parthenocarpic fruits, e.g., Banana. Parthenocarpy can be induced through the application of growth hormones like Auxins.
The first stimulus for fruit development comes from pollination while second stimulus is received from developing seeds and the third stimulus is provided by the availability of nutrients.
Advantage of seeds to Angiosperms
Seeds have better adaptive strategies for dispersal to new habitats and help the species to colonize in other areas. As they have sufficient food reserves, young seedling are nourished until they are capable of photosynthesizing on their own.
The hard seed-coat provide protection to young embryo. Being product of sexual reproduction, they generate new genetic combinations leading to variations.
Seed forms basis of our agriculture. Dehydration and dormancy of mature seeds are crucial for storage of seeds which can be used as a food throughout the year and also to raise crops in next season. Seed viability varies from species to species. '
Oldest viable seeds of lupine, Lupinus arcticus were excavated from Arctic Tundra. The seed germinated and flowered after an estimated record of 10,000 years of dormancy.
A recent record of 2000 years old viable seed is of date palm (Phoenix dactylifera), discovered during the archeological excavation at King Herod's Palace near Dead Sea.
The ability of a seed to retain the power of germination is called viability. It can be tested by using 0.1 % solution of triphenyl tetrazolium chloride (TTC).
Orchid fruits contain thousands of tiny seeds. Similar in the case in fruits of Ficus and parasitic species such as Orobanche and Striga.
apomixis and polyembryony
- Books Name
- A TEXT OF BIOLOGY - CLASS XII
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Biology
APOMIXIS AND POLYEMBRYONY
Apomixis is the formation of new individuals directly through asexual reproduction without involving the formation and fusion of gametes.
It is of two types :
(i) Agamospermy and
(ii) Vegetative Propagation
Agamospermy is a mode of apomixis in which seeds are formed but are asexual in nature as the embryo develops directly without gametic fusion.
There are several types of agamospermy :
(i) Adventitive Embryony (Sporophytic budding) : Embryo arises from diploid sporophytic cells such as nucellus or integuments (other than egg), e.g., Citrus, Opuntia, Mango.
(ii) Recurrent Agamospermy : In this method, a diploid embryo sac is formed from megaspore mother cell which has a diploid egg or oosphere. The diploid egg grows parthenogenetically into diploid embryo, e.g., Apple, Pear, Allium. Diploid embryo sac can develop directly from either the diploid megaspore mother cell (diplospory) or diploid nuclear cell (apospory).
(iii) Non-recurrent Agamospermy : Embryo develops parthenogenetically from haploid egg, e.g., Banana.
(iv) Apogamy: It is formation of sporophyte or embryo directly from cells of gametophyte. In higher plants, only diploid apogamy is successful. In lower plants, haploid apogamy is equally successful.
Polyembryony is the phenomenon of having more than one embryo in a seed, discovered in Citrus by Leeuwenhock. There may be more than one egg cell in an embryo sac or more than one embryo sac in an ovule.
All the egg cells may get fertilised. Synergids and antipodal cells may also form embryos (mixed polyembryony). Occurrence of polyembryony due to fertilisation of more than one egg is called simple polyembryony.
Formation of extra embryos through sporophytic budding is called adventitive polyembryony.
Polyembryony is quite common in onion, groundnut, mango, lemon and orange. In some of these cases stimulus of pollination may be required. Citrus seed has 2-40 embryos, one normal and the rest adventitive mostly nucellar.
Importance of Apomixis:
Hybrid varieties of several of our food and vegetable crops are being extensively cultivated because they provide higher and better yield. However, there is one major drawback.
Hybrid seeds have to be produced every year because seeds collected from hybrid plants, if sown subsequently, do not maintain hybrid characters due to segregation of traits.
Production of hybrid seeds every year is costly thus increasing the cost of crop production. This can be avoided if apomixis is introduced in hybrid seeds.
Concept Builder
(i) The study of seed is called spermology.
(ii) Chalazosperm is perisperm like tissue in chalazal region. It is a substitute for endosperm e.g. Cynastrum.
(iii) Seed viability in Oxalis -few days
(iv) Apomixis is common in some species of Asteraceae and grasses.

 ACME SMART PUBLICATION
ACME SMART PUBLICATION
