- Books Name
- Kaysons Academy Chemistry Foundation Book
- Publication
- Kaysons Publication
- Course
- JEE
- Subject
- Chemistry
Suspension and Evaporation
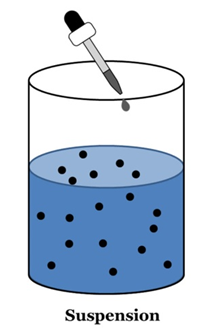
Suspension
A suspension is a heterogeneous mixture in which the solute particles do not dissolve but remain suspended throughout the bulk of the medium.
Properties of a Suspension
- Suspension is a heterogeneous mixture.
- Its particles can be seen by naked eyes.
- Its particles scatter a beam of light passing and make its path visible (Tyndall effect).
- It is unstable. The soluble particles settle suspension is left undisturbed or they can by the process of filtration.
Colloidal Solution

A colloid (or colloidal solution) is a mixture that is heterogeneous but appears to be homogeneous as the articles are uniformly spread
throughout the solution. e.g., milk, shaving cream, cheese etc.
Properties of a Colloid
- A colloid appears to be homogeneous but actually it is heterogeneous.
- A colloid appears to be homogeneous but actually it is heterogeneous.
- The size of particles of colloid is very small. They can not be seen even with a microscope.
- Its particles can pass through filter paper, therefore, a colloid cannot be separated by filtration. However, they get separated by a special technique, called centrifugation.
Some Common Examples of Colloids
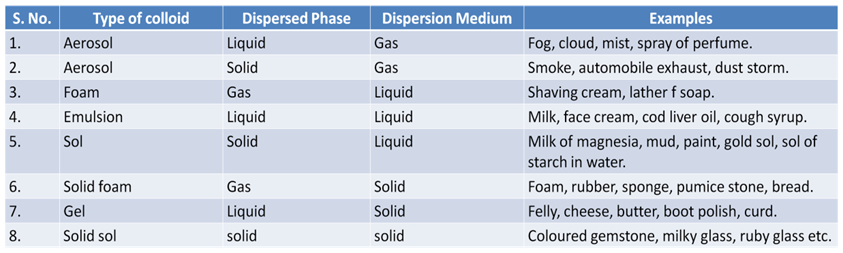
Components of a Colloid
The solute like component form the dispersed phase and the component in which the dispersed phase is suspended is known as the dispersion medium.
Evaporation
This method can be used to separate a volatile component (solvent) from a non-volatile component of a mixture.
Centrifugation
(Separation of Cream from Milk)
Two components having difference in densities can be separated by centrifugation.
Illustration
How the separation of cream from milk takes place?
Solution
To separate cream from milk, milk is churned for 2-3 minutes. Cream collects at the centre and being lighter than milk floats at the top of the mixture.
Decantation
This method is also applied where a difference between densities of components lies. A less soluble solid from a liquid can be separated by first keeping the mixture undisturbed and then decanting the liquid slowly leaving the solid in the first container.
Separating Funnel
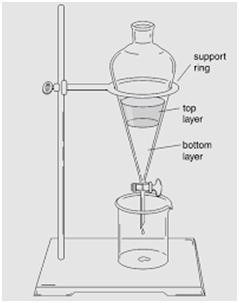
(Separation of Mixture of Two Immiscible Liquids)
This method is used for separating two immiscible liquids and is also based on the difference in densities.
Illustration
Name the apparatus used to separate these two liquids.
Solution
Separating funnel in used to separate oil from water.
Illustration
State the principle involved in this process.
Solution
This process is based upon the principle that immiscible liquids are separated out in layers depending upon their densities.
Illustration
How the layer is formed in separating funnel?
Solution
Kerosene oil being lighter will form the upper layer while water being heavier will form the lower layer.
Illustration
A mixture containing two liquids is placed in separating funnel. Answer the following.
- What type of liquids form the mixture?
- Which of the liquids will form the lower layer?
- What is the basis of this method?
Solution
- Two liquids which are immiscible with each other form the mixture.
- The heavier liquid will form the lower layer.
- The method is based on
(a):- mutual immiscibility of the two liquids
(b):- difference in the densities of the two liquids.

 Kaysons Publication
Kaysons Publication
