- Books Name
- Class-8 Science Book
- Publication
- PathSet Publications
- Course
- CBSE Class 8
- Subject
- Science
Physical Properties of Metals and Non-Metals
Elements can be classified into the following two groups depending on their physical and chemical properties:
- Metals
- Non-metals
Metals
Metals are opaque and lustrous elements that are good conductors of heat and electricity.
Example: Iron, copper, zinc.
- The elements which are hard, shiny can be beaten into sheets, drawn into wires and are good conductors of heat and electricity are generally metals. For example iron, copper, gold, etc.
- In nature, most metals occur in the combined state as minerals and they are reactive.
- Only a few unreactive metals like gold, silver, platinum are found as free metals in the earth’s crust.
- Minerals from which metals can be profitably extracted are called ores. For example, calcium occurs in limestone (calcium carbonate) or iron in the ore Haematite.
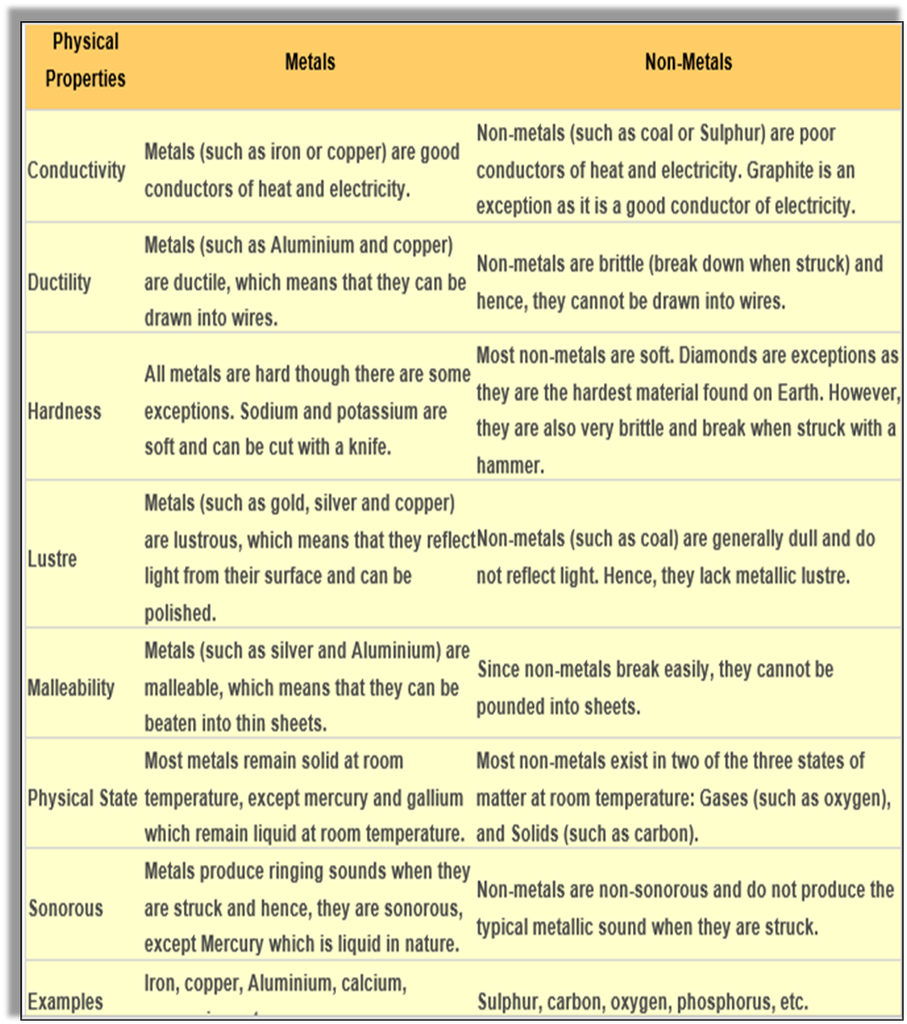
Physical properties of metals:
Malleable – ability to be beaten into sheets. The property by virtue of which metal can be beaten into sheets is called malleability. Eg- We use Aluminum foil to pack food. Aluminum and silver foil.
Ductile – ability to be drawn into wires. The property by which metals can be drawn into wires is called ductility. Metals like copper, silver and Aluminum can be drawn into wires.
Lustrous – ability to shine. Metals such as gold, silver and copper all have lustre, that is they have an ability to shine and reflect light. Therefore, they are lustrous.
Sonorous – makes a ringing sound when strikes with a hard surface, e.g. bells.
Hard in nature -e.g. iron, lead
Exception – (Sodium and potassium are soft metals and can be cut with a knife).
Good conductors of heat and electricity. They are good conductors of heat and electricity. Copper is the best conductor of electricity followed by gold and Aluminum.
Mostly solids Exception – (Mercury is the only liquid metal).
Mercury - is the only metal that is found in the liquid state at room temperature.
Metals like Sodium and Potassium are soft and can be cut with a knife.
High melting and boiling point -With the exceptions of sodium, potassium and mercury most of the metals have high melting and boiling point.
High Densities -Metals have high densities.
High tensile strength - Most metals have high tensile strength.
Non- Metals
The elements that do not have the properties of metals are known as non-metals. Non-metals are widely present in the earth’s crust and atmosphere. All oceans and water bodies have an abundance of these elements. Examples: Carbon, hydrogen, and oxygen.
- The elements which are brittle, dull cannot be beaten into sheets or drawn into wires and are poor conductors of heat and electricity are generally non-metals.
- For example, oxygen and nitrogen occur in a free state in the air and in the combined state in earth’s crust. Sulfur occurs both in the free and the combined state in the earth’s crust.
- The noble gases, helium, neon, argon, krypton, xenon occur only in Free State.
Physical properties of Non-Metals:
▪ Non-malleable – they are not malleable cannot be made into sheets e.g. Sulphur breaks into powdery mass when beaten with a hammer.
▪ Non-ductile – Non-metals are brittle that is they are not ductile. Hence, cannot be made into wires.
▪ Non-lustrous/dull - Non-metals do not have lustre except iodine and graphite. e.g. coal.
▪ Non-sonorous – cannot make a ringing sound when struck with any object.
▪ Soft, break easily, e.g. Sulphur, Pencil lead.
▪ Poor conductors of heat and electricity, e.g. phosphorus
▪ Exception – (Graphite is a good conductor of heat and electricity).
▪ Non-metals are gases or solid at room temperature, except bromine which is liquid at room temperature. It can be solid (Sulphur), liquid (bromine), or gaseous (oxygen). They usually have low densities. Non-metals have low melting points and boiling points.
Physical Properties of Metals and Non-metals
Note: Copper Vessels also acquire a dull green coating (made up of copper hydroxide and copper carbonate) when they are exposed to moist air. It is called Verdigris.

 PathSet Publications
PathSet Publications
