- Books Name
- CBSE Class 6 Science Book
- Publication
- Param Publication
- Course
- CBSE Class 6
- Subject
- Science
SEPARATION OF SALT FROM SEA WATER
Sea water contains many salts mixed in it. One of these salts is the common salt. When sea water is allowed to stand in shallow pits, water gets heated by sunlight and slowly turns into water vapour, through evaporation. In a few days, the water evaporates completely leaving behind the solid salts. Common salt is then obtained from this mixture of salts by further purification.

Obtaining salt from sea water
USE OF MORE THAN ONE METHOD OF SEPARATION :
Often, one method is not sufficient to separate the different substances present in a mixture. In such a situation, we need to use more than one of these methods.
For example :
It we take a mixture of sand and salt. Handpicking would not be a practical method for separating these. In order to separate them we keep this mixture in a beaker and add some water to it and leave it aside for some time. We will see the sand settling down at the bottom. The sand can be separated by decantation or filtration.
Now, we need to separate salt and water from the decanted liquid. For this we transfer this liquid to a kettle and close its lid. The kettle is heated for some time. We notice steam coming out from the spout of the kettle.
Then we hold a metal plate with some ice on it just above the spout of the kettle. Let all the water in the kettle boil off.
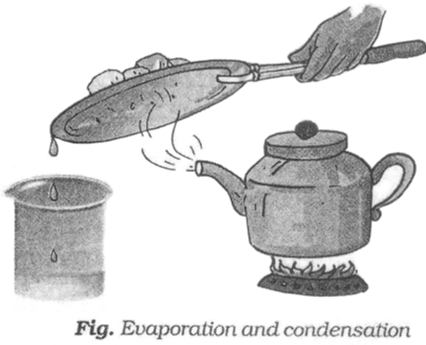
When the steam comes in the contact with the metal plate cooled with ice, it condenses and forms liquid water. The process of conversion of water vapour into its liquid form is called condensation.
After all the water has evaporated, salt is left behind in the kettle.
We have thus, separated salt, sand and water using processes of decantation, filtration, evaporation and condensation.

 Param Publication
Param Publication
