- Books Name
- Science Made Easy Science Book
- Publication
- Science Made Easy
- Course
- CBSE Class 9
- Subject
- Science
Atomic Models
→ From the knowledge of existence of subatomic particles like electron, proton and neutron in
atom, various atomic models were proposed by different scientists.
• Some of the atomic models:
(i) Thomson’s Model of Atom
(ii) Rutherford’s Model of Atom
(iii) Bohr’s Model of Atom
→ The most trusted and scientifically established model of atom which is adopted these days
‘Quantum Mechanical Model of Atom’. It will be dealt in higher classes.
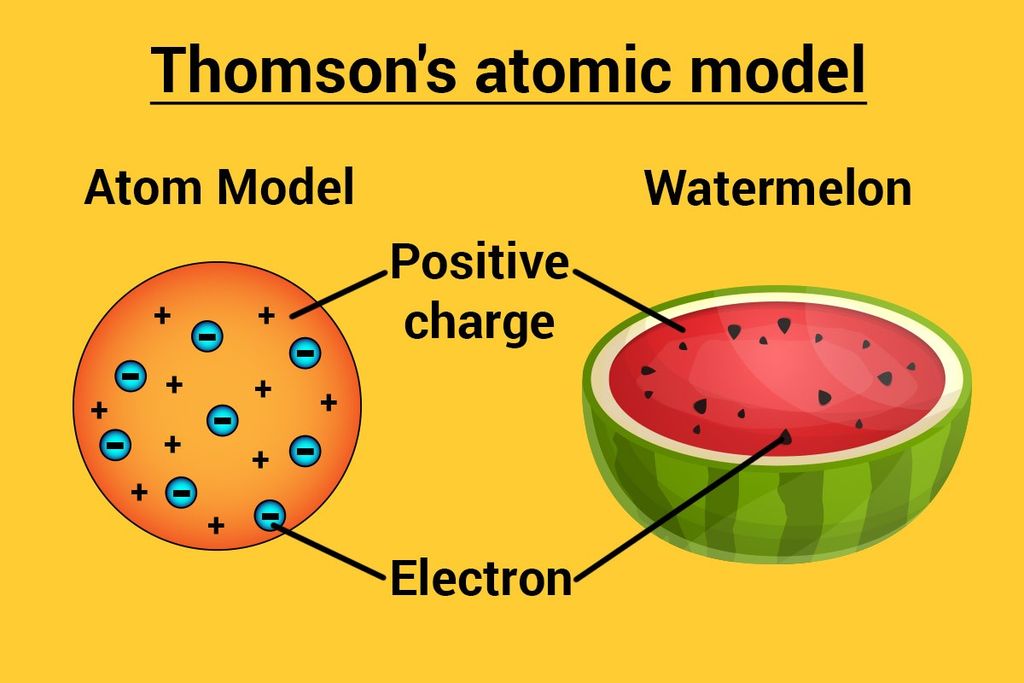
Thomson’s Atomic Model
→ This model is often called the ‘Water Melon Model’.
→ In this model, Thomson predicted the presence of electrons inside positive sphere (made up
protons), just same as seeds of watermelon are embedded in red edible part of watermelon.
→ Although this model explained neutrality of atom but couldn’t able to explain other scientific
experiments conducted on atom. Hence it was discarded.

 Vaishnav Publication
Vaishnav Publication
 ACERISE INDIA
ACERISE INDIA
