- Books Name
- Class-8 Science Book
- Publication
- PathSet Publications
- Course
- CBSE Class 8
- Subject
- Science
What is Combustion?
Combustion: It is a chemical process in which a substance reacts with oxygen to give off heat is called combustion.
In other words: Combustion is a chemical process in which a substance reacts with oxygen and generates heat in the process.

Any material or substance that undergoes combustion is known as a combustible substance. It is also called fuel.
Combustible substances are those substances that undergo combustion. It means that these substances give off heat and sometimes light (as a flame or glow) when they react with oxygen. Examples - Petrol, Diesel, etc. The fuel may be in a solid, liquid, or gas state. Sometimes, light is also given off during combustion, either as a flame or as a glow.
Inflammable Substances:
Those materials which have low ignition temperature and catch fire easily are termed inflammable substances. They burn with a flame. Example - Petrol, LPG, Alcohol etc.
Why do we say that food is fuel for our bodies?
When we eat food, it gets broken down into simpler substances that react with oxygen and generate energy (or heat). Hence, food is referred to as ‘fuel’ for our bodies.
Conditions necessary for the Combustion to take place are:
Combustion requires fuel.
Combustion only takes place when there is a fuel that produces heat and light when it catches fire. For example, petrol and diesel.
Combustion requires air.
We can prove it with the help of the following experiment: A candle keeps burning in the open air. However, when we cover it with a glass or a jar, it only burns until the oxygen inside it is consumed. Once the oxygen is exhausted, the flame of the candle flickers off.
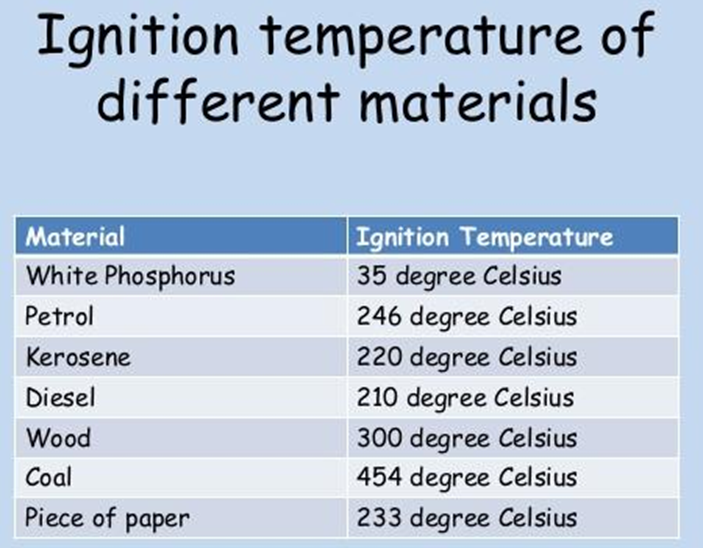
Combustion requires heat. The substance must reach its ignition temperature to catch fire. Ignition Temperature: the minimum temperature at which any material catches fire is known as ignition temperature.
Example-
- If the temperature of a combustible substance is lower than the ignition temperature, then the substance will not burn.
- Cooking oil catches fire when a frying pan is kept for long on a burning stove.
- Kerosene oil and wood do not catch fire on their own at room temperature. But, if kerosene oil is heated a little, it will catch fire. But if the wood is heated a little, it would still not catch fire.
Matchsticks
Since ages, matchsticks are in use. Long ago, Egyptians used small pieces of pinewood dipped in Sulphur as matches. These days matchsticks are a lot safer. Modern matchsticks are made up of a mixture of antimony trisulphide and potassium chlorate with some glue and starch applied to the head of the match. The rubbing surface has powdered glass and some red phosphorous. On the striking match against a rough surface, red phosphorous gets converted into white phosphorous and it reacts with potassium chlorate to ignite antimony trisulphate and so the combustion takes place
THREE ESSENTIAL REQUIREMENTS FOR PRODUCING FIRE
- Fuel
- Air (With Oxygen in it).
- Temperature above the Ignition temperature
Ignition temperature is the lowest temperature at which a substance catches fire or starts burning.

 PathSet Publications
PathSet Publications
