- Books Name
- CBSE Class 7 Science Book
- Publication
- Param Publication
- Course
- CBSE Class 7
- Subject
- Science
Physical Change
The properties such as size, shape colour and state of a substance are called its physical properties.
When a substance undergoes a change in its physical properties, that change is said to be physical change. During a physical change, no new substance is formed. Physical changes are generally reversible changes.
Remember
• Reversible change:
A change in which we can get back the initial substance by reversing the action is an reversible change.
Activity–1
To observe a physical change with the help the of ice cubes. 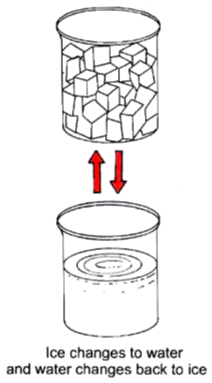
Procedure :
• Take a few ice cubes in a beaker.
• Keep them in the open for 4–5 minutes.
Now ,we will observe that ice (solid) changes into water (liquid). There is a change in the state of ice from soild to liquid.
• Now pour this water back into the ice tray and keep it in the freezer for 30 minutes.
Now, we will get back the ice. Therefore, it is a physical change.
Activity–2
To observe a physical change with the help of a sugar solution.
Procedure : 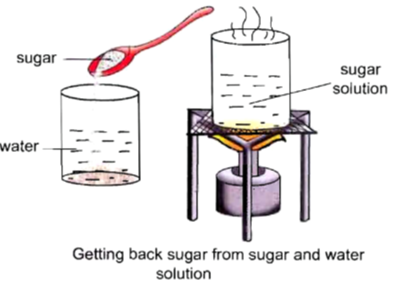
• Take 100 mL of water in a beaker.
• Dissolve a spoonful of sugar into it.
• Now, we will get a solution of sugar and water.
• Now, heat this solution over a burner for sometime.
• Now, we will observe that slowly the water evaporates and sugar is left at the solution.
• We will be able to get back sugar. Therefore, it is a physical change.
Characteristics of physical change
(i) No new or different product is formed : The composition of molecules of the substance remains unaltered.
Example : Ice melts to form water. In this example only the appearance (state) of matter has changed from solid to liquid. However, the composition of the molecules of ice or water remains same, i.e., for every 1 g of hydrogen, 8 g of oxygen is required . Thus, only a physical change has occurred.
(ii) The change is temporary and is usually reversible : It means the change can be reversed by altering the causes which produce the change.
Example : The water formed from ice can be changed back to ice by placing it in a freezing mixture (a mixture of ice and common salt).
(iii) There is no change in the weight of substance : During a physical change it is only the energy which is added or removed. No matter is added during a physical change. Similarly, no matter is removed during a physical change. Therefore, mass of the substance remains the same.
Some Examples Involving Physical Changes :
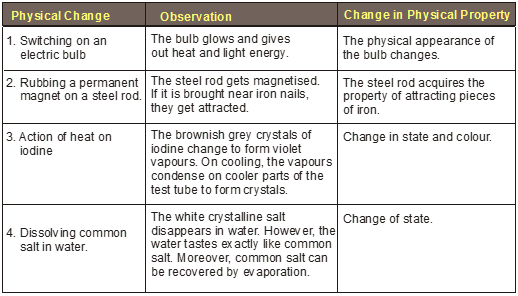
Some Common Examples of Physical Changes
• Formation of dew.
• Evaporation of water.
• Crystallisation of sugar from its solution.
• Ringing of an electric bell.
• Breaking of a glass pane.
• Freezing of ice cream.
• A rock rolling down a hill.
• Bending of a glass tube by heating.
• Melting of wax.
• Sublimation of camphor.
Chapter 6: Physical and Chemical Changes

Matter is capable of undergoing changes, which are classified as either physical or chemical. Physical changes in matter are reversible: An ice cube can melt into liquid water, and then the liquid water can be frozen back into an ice cube. Chemical changes, on the other hand, are not reversible: A log burned in a fire turns to ashes, but the ashes cannot be changed back into a log
Physical changes
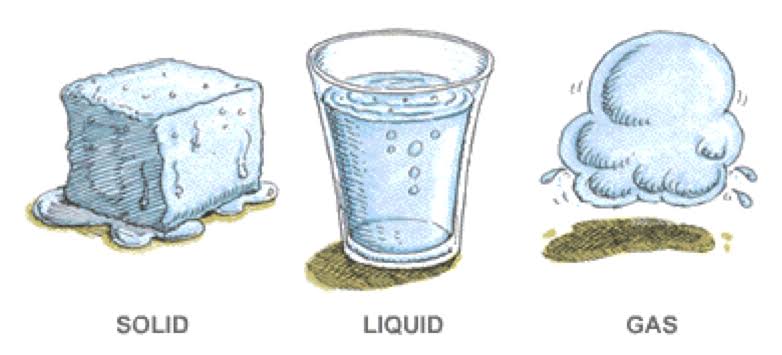
In a physical change, the material involved in the change is structurally the same before and after the change. Types of some physical changes are texture, shape, temperature, and a change in the state of matter. A change in the texture of a substance is a change in the way it feels. For instance, a block of wood may feel rough when you run your finger across it but rubbing the wood with sandpaper smooths the surface so it no longer feels rough. The wood itself has not changed during sanding to become a new material, only the texture of the surface changed. A piece of metal may be heated in a fire until it glows, but the metal is the same material before heating and after cooling. Similarly, when a material changes phase, it only changes physically; the substance is still the same.

 Param Publication
Param Publication
 Grow Career Publication
Grow Career Publication
