- Books Name
- CBSE Class 6 Science Book
- Publication
- Param Publication
- Course
- CBSE Class 6
- Subject
- Science
Properties of Materials
We generally use a tumbler to keep a liquid. A tumbler cannot be made from cloth. We need a tumbler made from glass, plastics, metal or other such material that will hold water. Similarly, it would not be wise to use paper-like materials for cooking vessels.

Using a cloth tumbler
we choose a material to make an object depending on its properties, and the purpose for which the object is to be used.
Some properties are discussed here.
(i) Appearance :
Materials usually look different from each other. Wood looks very different from iron. Iron appears different from copper or aluminium. At the same time, there may be some similarities between iron, copper and aluminium that are not there in wood.
Activity–2
AIM : To show that certain materials have lustre.
Material required : (i) Small pieces of paper (ii) cardboard (iii) a thin piece of wood (iv) a small strip of plastic (v) thin wires of copper, aluminium and iron (vi) heavy scissors or a metal cutter
(vii) sand paper.
Method :
(a) Cut each of the materials with heavy scissors or metal cutter. Carefully observe the cut surface of each material.
We will notice that in case of paper card board, wood plastic, etc., the cut surface is not shining.
However, in case of thin wires of copper, aluminium and iron, the cut surface is shining.
(b) Rub each of the material with sand paper. Carefully observe the sand papered surface of the material.
We will notice that in case of paper cardboard, plastic and wool, the sand papered surface is not shining.
However, in case of thin wires of copper, aluminium and iron, the sand papered surface is shining.
So it is cleared that certain materials like metals have lustre.
(ii) Hardness :
When we press different materials with our hands, some of them may be hard to compress while others can be easily compressed.
Materials which can be compressed or stretched easily are called soft while some other materials which are difficult to compress are called hard. For example, cotton or sponge is soft while iron is hard.
(iii) Solubility :
Some substances like sugar, salt etc. completely disappear or dissolve in water. These substances are soluble in water. Other substances like sand, chalk powder etc. do not mix with water and do not disappear even after we stir for long time. These substances are insoluble in water.
Water plays an important role in the functioning of our body because it can dissolve a large number of substances.
- Books Name
- Class 6 Science Book
- Publication
- PathSet Publications
- Course
- CBSE Class 6
- Subject
- Science
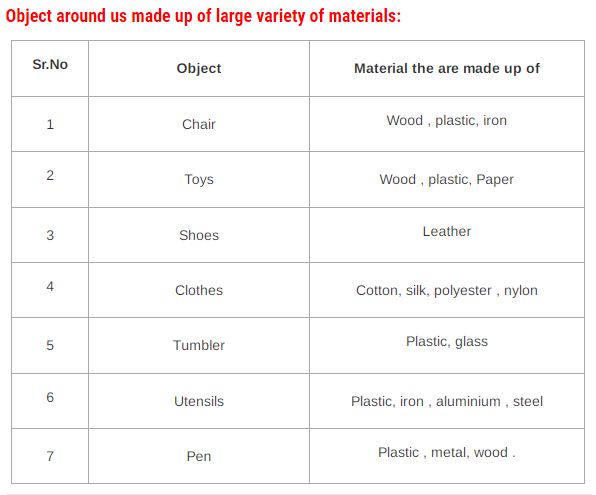
OBJECTS AROUND US

When we look around, we find ourselves surrounded by a number of objects. Some of these different objects are made from a number of different materials, while others are made using the same material. For Example, both desk and chair are made from wood while pen and dustbins are made using plastic. The material from which an object is made depends on its properties.
OBJECT – Object is a thing that is made up of one or more material.
Example – Pencil, chair, Table, Book, Food Item etc.
There is a vast variety of objects everywhere.
All objects around us are made up of one or more materials.
A thing can be made of different materials.
Many things can be made from the same material.
Material – Object is made up of Material.
Example – Pencil is an Object and it is made up of Materials like wood, metal, Graphite.
Classification: The process of sorting and grouping things according to some basis is called classification.
Why do we need to group objects?
We need to group objects for a number of reasons:
- Convenience to store: We often group objects in order to store similar objects together in order to make locating them easier in the future. Even in our homes, we store spices together in the kitchen while storing washing products in our bathrooms.
- Convenience to study: We also group objects so that it becomes easy for us to study their features as well as the patterns of these features.
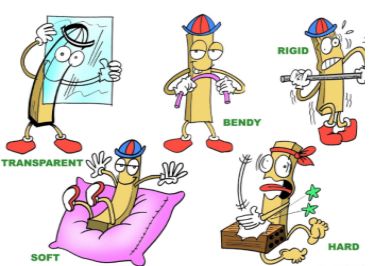
Basis of grouping: We can group materials on the basis of the similarities and differences in their properties. Each material has its own properties, that may differ from others. However, some properties may be common to one or more materials.
Grouping On The Basis Of Common Properties:
Objects are made of different materials. One material can be used to make different objects. This is possible because different types of materials have different properties. We have to choose materials with the right properties based on what we want to use it for. For example, a chalk made of wood or plastic would be of no use because it cannot be used to write on the blackboard.
Materials have different properties like roughness, lustre, transparency, solubility, flotation, attraction towards a magnet, conduction of heat, and conduction of electricity. Let us study these properties one by one.

 Param Publication
Param Publication
 PathSet Publications
PathSet Publications
