- Books Name
- ACME SMART COACHING Chemistry Book
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 12
- Subject
- Chemistry
Proteins
Proteins : Proteins are the most abundant-biomolecules of the living beings. The chief source of proteins are milk, cheese, pulses peanuts, fish meat etc. These are high molecular mass complex, biopolymers of amino acids. The word protein is derived from Greek word proteios which means primary or of prime importance.
Amino Acids : Each living cell is made up of thousands of different proteins All proteins contain the elements like carbon, hydrogen, oxygen, nitrogen and sulphur. Some of these also contain phosphorus, iodine and traces of metals such as, Fe, Cu, Zn, & Mn. All proteins are polymers of a-amino acids and on partial hydrolysis give peptides of varying molecular masses which upon complete hydrolysis give a-amino acids.
![]()
Type of a-amino acids :
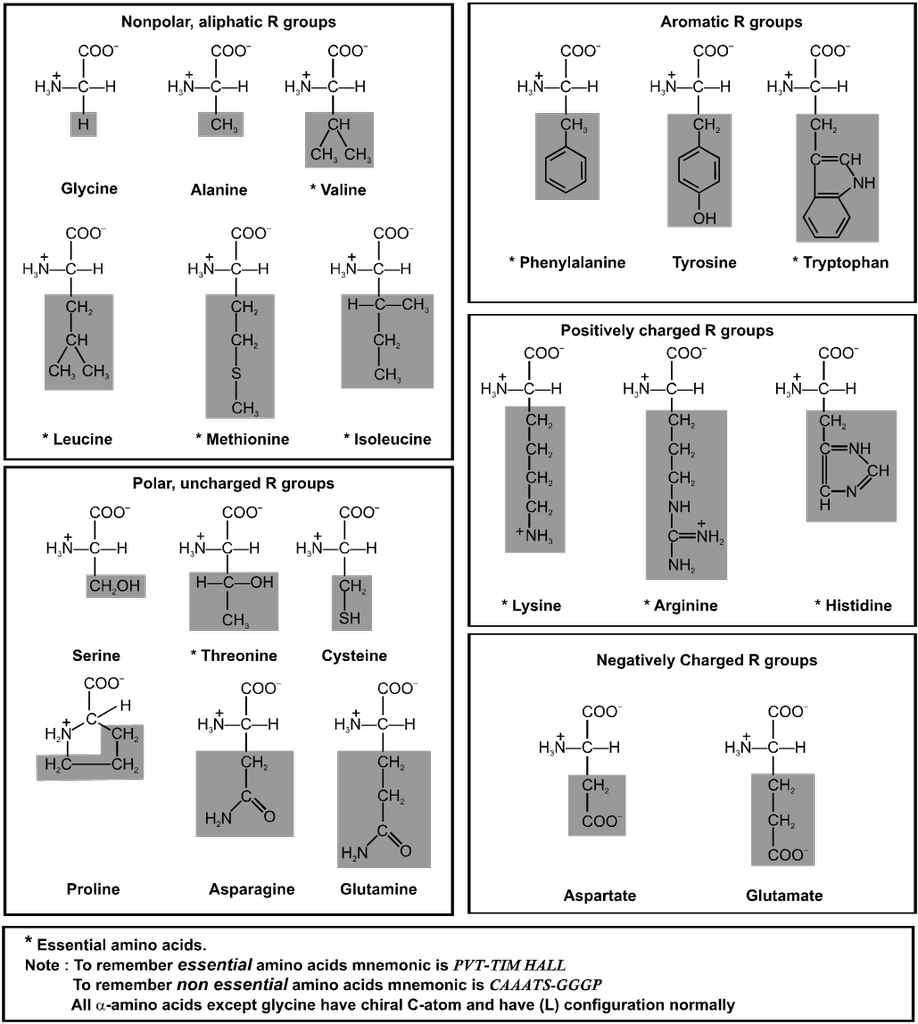
These organic compounds contain amino as well as carboxylic acid group. On the basis of position of amino group in the chain, these are named as a, b, g etc. amino acid.
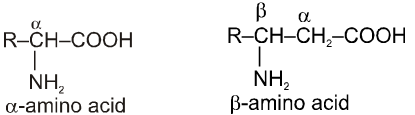
Amino Acid : Building Blocks of Proteins
Amino acids are the building blocks of the molecular structure of the important and very complex class of compounds known as proteins. The proteins on hydrolysis yield mixture of the component amino acids.
Amino acids are bifunctional compounds containing both an amino and a carboxylic acid group. They are represented by the general formula :
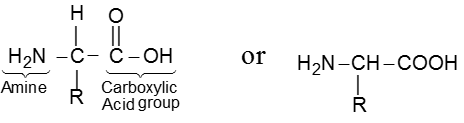
where, R = alkyl, aryl, or any other group.
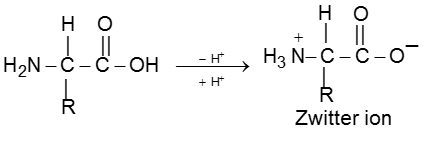
Zwitter ion (Dipolar Nature of Amino acids) :
In a neutral amino acid solution, the –COOH loses a proton and the –of the same molecule picks up one.The resulting ion is dipolar, charged but overall electrically neutral. This is called Zwitterion (German, “two ions”). Therefore amino acids are amphoteric.
There are around 20 amino acids in the living system.
Classification of Amino Acids :
(A) On the basis of synthesis :
(i) Non essential amino acids : 10 amino acids are synthesis in our body and these are said to be non essential amino acids (eg. Gly, Ala, Glu, Asp, Pro and Cys).
(ii) Essential amino acid : 10 amino acids which are necessarily be present in our diet are called essential amino acids (eg. Val, Leu, I leu, Lys and Phe).
(B) On the basis of functional groups present :
(i) Neutral amino acids : If only one – NH2 and one – COOH groups are present. eg. Glycine, Alanine, Valine, Leucine etc.
(ii) Acidic amino acid : If one basic and two acidic groups are present. Additional acidic functional group must be present in the side chain. E.g. Aspartic acid and Glutamic acid.
(iii) Base amino acid : If two basic and one acidic group is present. Additional basic group must be present in the side chain. E.g. Arginine, Lysine & Histidine.
Isoelectric point of a-amino acids :
When an ionised amino acid is placed in an electric field, it will actually migrate towards the opposite electrode. Depending upon the pH of the medium, three things can happen. The positive form (II) will migrate to the cathode, the neutral form (I) (Zwitter ion) will not migrate, while the negative form (III) will migrate to the anode. The pH at which the amino acid shows no tendency to migrate when placed in an electric field is known as its isoelectric point. This is characteristic of a given amino acid. Thus glycine has its isoelectric point at pH 6.1.
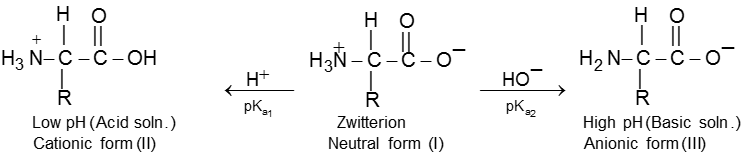
Isoelectric Point : The pH at which the amino acid shows no tendency to migrate when placed in an electric field is known as its isoelectric point.
Because of amphoteric nature in acidic solution it exist as the +ve ion. Hence it migrate towards cathode while in basic solution it exist as –ve ion and migrates towards anode.
In strongly acidic medium
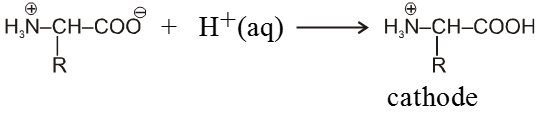
In strongly basic medium
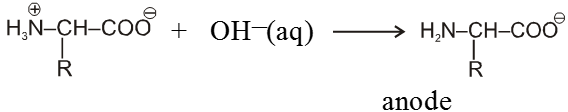
At some intermediate pH amino acids exist as a neutral dipolar ion i.e. the concentration of the cation and anions are equal and it does not migrate towards either electrode, this pH is called iso electric point of amino acid which is different for different amino acids.
For example :
(i) For neutral amino acid : pH of isoelectric point varies between 5.1 to 6.5. e.g. Glycine has pH value 6.0.
PI for neutral amino acid is calculated as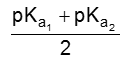
(ii) For acidic amino acid : Where there are two COOH groups and one NH2 group then isoelectric pH is around 3. e.g. Aspartic acid.
Aspartic acid :
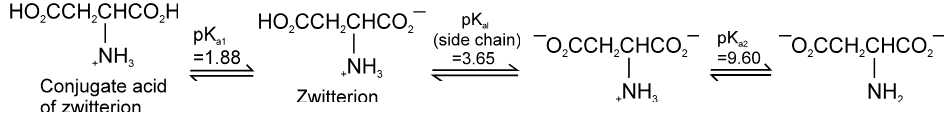
PI for acidic amino acid is calculated as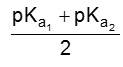
The pI of aspartic acid is the average of pKa1 (1.88) and the pKa of the side chain (3.65) or 2.77.
(iii) For basic amino acid : where there are two NH2 groups and one COOH group then isoelectric point varies between 7.6 to 10.8.
e.g. Lysine (9.8)
Lysine :
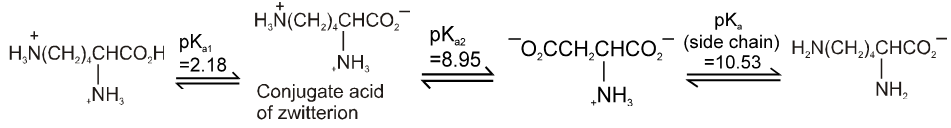
PI for basic amino acid is calculated as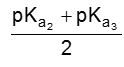
The PI of lysine is the average of pKa2 (8.95) and the pKa of the side chain (10.53) or 9.74.
Note : Amino acid has minimum aqueous solubilities at their isoelectric points.
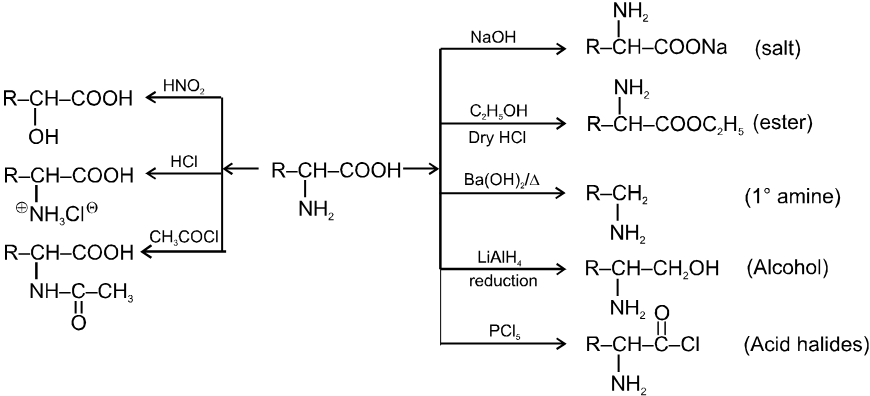
(a) General methods of preparation
1. Aminolysis of a-halocarboxylic acid
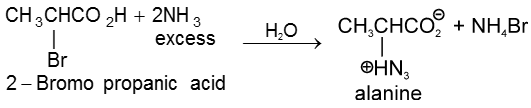
2. By strecker synthesis : Aldehyde reacts with a mixture of NH4Cl and NaCN to form a-aminonitrile (as an intermediate) which on hydrolysis gives an amino carboxylic acid.
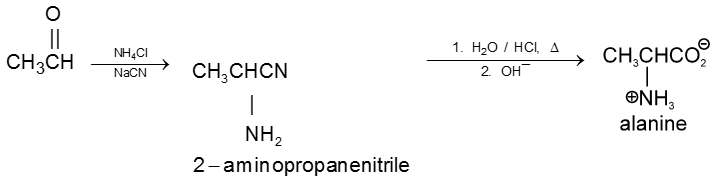
3. From aceto acetic ester :
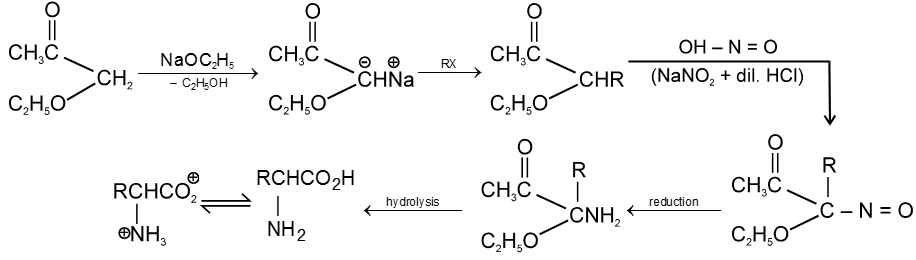
4. By Gabriel Synthesis :

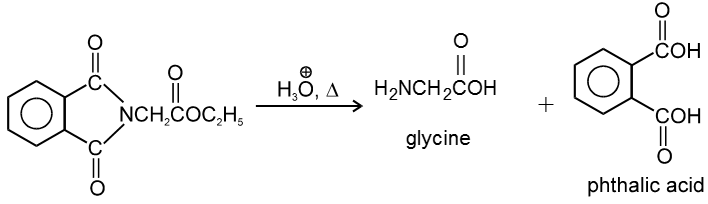
5. Effect of Heat :
a-amino acids undergo intermolecular dehydration on heating at about 200°C to give diketopiperazines.
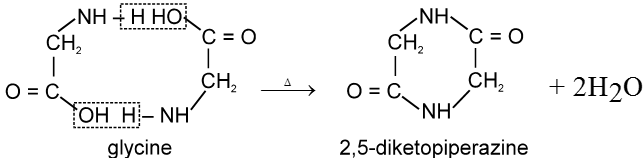
b-amino acids undergo intramolecular deamination on heating to form a, b-unsaturated acids.
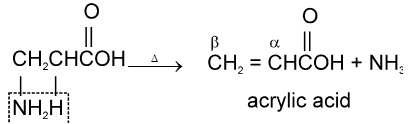
g-amino acids and d-amino acid undergo intramolecular dehyderation to form cyclic amides called.
Lactams.
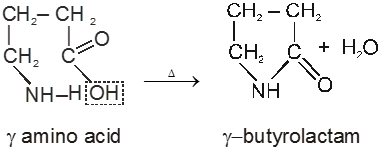
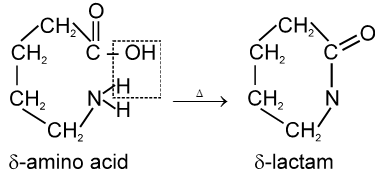
In case of d-amino acid, intramolecular cyclisation would given a seven-membered ring, which is formed with difficulty. Hence, there is intermolecular polymerisation forming nylon-6.
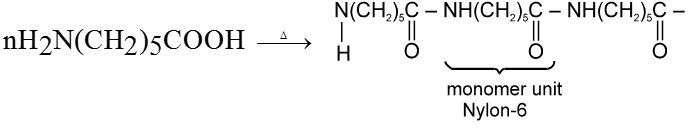
(b) Other Reactions of a-Amino Acid :
Peptide linkage :
Amino acid units are linked together by peptide ![]() – linkage and form polypeptides.
– linkage and form polypeptides.
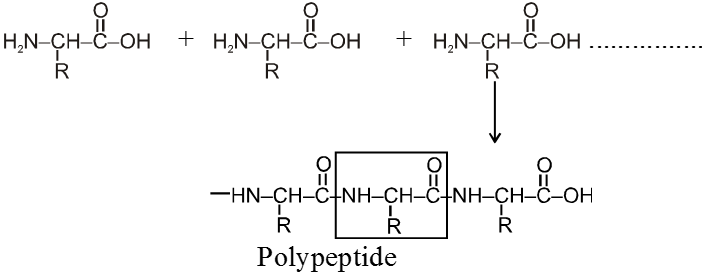
Note : Formation of protein linking together from different amino acids is not a random process. Each molecule of a given protein have the same sequence of amino acid along with its polypeptide chain. This tells about its own specific properties. Even the change of just one amino acid can change drastically the properties of entire protein molecules.
e.g. hemoglobin has 574 amino acids in definite sequence. The change of only one amino acid makes it defective hemoglobin which looses its property of carrying oxygen in blood stream and result a disease called sickel cell animea.
Writing and Naming of Polypeptides :
The structure of polypeptides are written in such a way that amino acid with free –NH2 group is written on the left hand side (N-terminal) of the polypeptide chain, while the amino acid with the free carboxyl group is written on the right hand side (C-terminal) of the chain. Thus a tripeptide alanylglycylphenylalanine (Al, Gly, Phe) is represented as follows :
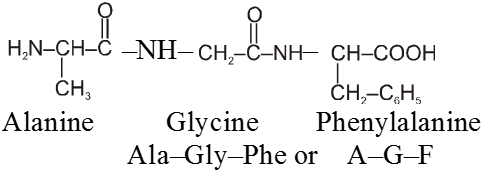
The name of any polypeptide is written starting from the N–terminal residue while writing the name the suffix “ine” in the name of the amino acid is replaced by “yl” (eg. glycyl for glycine, alanyl for alanine etc) for all the constituent amino acid except the C– terminal residue. This nomenclature is usually not used. Instead the three letter abbreviation or one letter codes for various a-amino acids present in the chain is used. For example the above tripeptide is named as Ala-Gly-Phe or A-G-F
Properties of polypeptides :-
(i) Polypeptides are amphoteric in character because of the presence of terminal ammonium and carboxylate ion as well as the ionised side chain of amino acid residues. Therefore like amino acids they neutralize both acids and bases and possess isoelectric points. At isoelectric point polypeptides have least solubility and hence can be separated.
(ii) The total contents of smaller peptides in tissues is small as compared to protein. However, like protein they also carry out many important functions in bio-system. Most of the toxins (poisonous substances) in animal and in plant sources are polypeptides. Some oligopeptides such as oxytoxin, vassopressin and angiotens are effective hormones while others act as sweetening agents. For example the dipeptide aspartyl phenyl alanine methyl ester called aspartame is 100-200 times more sweet than sucrose and is used as a suger substitute for dibetic patients
 Aspartame
Aspartame
Proteins :
Proteins are the most abundant-biomolecules of the living beings. The chief source of proteins are milk, cheese, pulses peanuts, fish meat etc. These are high molecular mass complex, biopolymers of amino acids. They occur in every part of the body and form fundamental basis of structures and determine functions of life. The word protein is derived from Greek word proteios which means primary or of prime importance.
Proteins are present in almost all the living cells of plant and animals. The protoplasm of plant and animal cells contains (10-20%) proteins. In human beings they are the main constituent of muscles, skin, hair, nail, tendons, arteries and connective tissue.
Classification of Proteins :
(I) On the basis of molecular structure :
(i) Fibrous proteins
(ii) Globular proteins
(i) Fibrous Proteins : When polypeptide chain run parallel and are held together by hydrogen and disulphide bonds then fiber like structure is formed such proteins are generally insoluble in water.
Ex. Keratin, myosine, collagen, etc.
(ii) Globular proteins : The polypeplide chain in these proteins is folded around it self to form spheroidal shape. The folding takes place in such a manner that lipophilic (non-polar, fat soluble) parts are pushed inwards and hydrophilic part (polar, water soluble) are pushed outwards. As a result water molecules interact strongly with the polar groups and hence globular proteins are soluble in water. As compare to fibrous proteins these are very sensitive to small changes in temperature and pH.
e.g. Insulin, albumin, thyoglobulin, antiadiodies, haemoglobin fibrinogen etc.
(II) On the basis of chemical composition :
(i) Simple proteins : These proteins on hydrolysis give only a-aminoacids. e.g. albumin in the white portion of eggs, glutenin in wheat, oxygenin in rice, keratin in hair, nails, horns etc.
(ii) Conjugated Proteins : These are the proteins in which protein part is combined with non-protein part. These proteins on hydrolysis give a non-protein in addition to the a-amino acids (Prosthetic group). The main function of prosthetic group is to control the biological function of the protein. Prosthetic groups are carbohydrate, phosphate group, lipids (ester of higher fatty acids) and so on. Casein of milk, haemoglobin of blood are conjugated proteins.
(iii) Derived Proteins : These are the degradation products obtained by partial hydrolysis of simple or conjugated proteins with acids, alkalies or enzymes. e.g. proteoses peptones, and polypeptides
Protein ® Proteoses ® Peptones ® Polypeptides
Structure of Proteins :
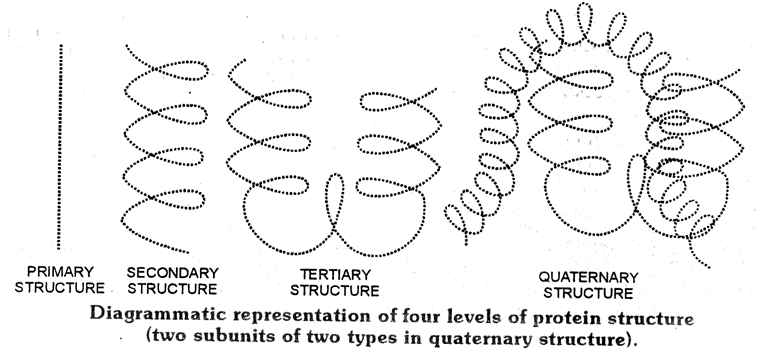
Proteins are biopolymers having a large number of amino acids through peptide bonds having three dimensional (3D) structures. Structure and shape of proteins can be studied and evaluated at four different levels i.e. primary, secondary, tertiary and quarternary, each level being more complex than the previous one.
(i) Primary structure of Proteins :
The manner and specific sequence in which different amino acids are joined to form polypeptides in a protein molecule is called 10 structure of that protein. Any change in this primary structure i.e sequence of amino acids creates a different proteins.
The first primary structure of a protein i.e INSULIN was determined by Frederic Sanger (a British chemist) and for this work he was awarded the Nobel prize in 1958. The different chemical and biological properties of various proteins are primarily due to the difference in their primary structures.
Normal Haemoglobin –Val – His – Leu – Thr – Pro – Glu –Lys–
Sickle cell anemia Val – His – Leu – Thr – Pro – Val – Lys–
(ii) Secondary structure of Proteins :
The conformation in which the polypeptides chains assume a shape as a result of H-bonding is called the secondary structure of protein.
Types:
(a) a-Helix structure
(b) b-pleated sheet structure
(iii)Tertiary structure of proteins :
The tertiary structure of protein represent overall folding of the polypeptide chains i.e further folding of the secondary structure. In other words, tertiary structure refers to the manner in which the entire protein molecule folds up in the three dimensional space to produce a specific shape. It gives rise to two major molecular shapes fibrous and globular. The main forces which stabilise the 2º and 3º structures of proteins are hydrogen bonds, disulphide linkages, vander waals and electrostatic forces of attraction.
(iv) Quarternary structure of proteins :
Although many proteins exist as a single polypeptide chain but there are certain proteins which are composed of two or more polypeptide chains referred to as sub-units or protomoss. These subunits may be identical or different and are held together by non-polar covalent forces such as, H-bonds, vanderwaals interaction and electrostatic interactions. The quarternary structure refers to the determination of the number of sub-units and their special arrangement w.r.t. each other in an aggregate protein molecule The Haemoglobin possesses quarternary structure and it is an aggregate of four polypeptide chains or sub-units.
A diagramatic representation of all four structure is given below, each ball represent an amino acid.
Denaturation of proteins :
A protein found in a biological sytem having a unique three dimensional structure and specific biological activity is called a native protein. When a protein in its native form is subjected to a physical change like change in temperature or pH, the hydrogen bonds are disturbed. As a result-globules unfold and helices get uncoiled and protein loses its biological activity. This is called Denaturation of proteins. During denaturation 2º and 3º structure are destroyed but 1º structure remain intact.
- Changing the pH denatures proteins because it changes the charges on many of the side chains. This disrupts electrostatic attractions and hydrogen bonds.
- Certain reagents such as urea and guanidine hydrochloride denature proteins by forming hydrogen bonds to the protein groups that are stronger than the hydrogen bonds formed between the groups.
- Detergents such as sodium dodecyl sulphate denature proteins by associating with the non polar groups of the protein, thus, interfering with the normal hydrophobic interactions.
- Organic solvents denature proteins by disrupting hydrophobic interactions.
- Proteins can also be denatured by heat or by agitation. Both increase molecular motion, which can disrupt the attractive forces. The coagulation of egg white on boiling and curdling of milk caused by the bacteria present in milk are common examples of denaturation of protein.
Renaturation of proteins :
The denaturation may be reversible or irreversible. The coagulation of egg white on boiling and curdling of milk are an example of irreversible protein denaturation. There is some cases where the process in actually reversible. The reverse process is called renaturation. In such cases when the temperature and pH of a denatured protein are brought back, the secondary and tertiary structures of protein are restored.
Test for proteins:
(a) Biuret test : Biuret reagent is a blue colour mixture of sodium hydroxide and copper sulphate. The blue colour of the reagent turns violet in the presence of proteins.
Condition :
- At least two peptide linkages must be present.
eg. Dipeptides do not give this test but tri, tetra,..........can give this test.
(b) Xanthoprotic:
Protein + conc. HNO3 ® Orange colour alkaline solution
(c) Ninhydrin test :
Proteins or Amino acids Purple coloured product.

 ACME SMART PUBLICATION
ACME SMART PUBLICATION
