- Books Name
- ACME SMART COACHING Biology Book
- Publication
- ACME SMART PUBLICATION
- Course
- CBSE Class 11
- Subject
- Biology
Protoplasm is a complex mixture of both organic and inorganic compounds.
Molecules found in the protoplasm of cells are called biomolecules.
A Comparison of Elements Present in Non-living and Living matter
The collection of various types of molecules in a cell is called the cellular pool.
The cellular pool consists of various types of biomolecules such as : (a) water (b) inorganic materials (c) organic compounds.
The small molecules of low molecular weight, simple molecular conformations and higher solubilities are called micromolecules.
These include minerals, water, amino acids, simple sugars and nucleotides.
The various minerals found in cells have many uses.
Mitochondria are rich in manganese.
Molybdenum is necessary for fixation of nitrogen catalysed by the enzyme nitrogenase.
Copper occurs in cytochrome oxidase.
Magnesium is essential for a large number of enzymes, particularly those utilising ATP.
Ca and Mg decrease the excitability of nerves and muscles.
(i) Sodium and potassium are responsible for the maintenance of extracellular and intracellular fluids through the osmotic effects of their concentration. These two ions are also responsible for the maintenance of membrane potential and transmission of electrical impulses in the nerve cells. Both in cells and in extracellular fluids, diabasic phosphate (HPO42–) and monobasic phosphate (H2PO42–) act as acidbase buffers to maintain the H+ ion concentration.
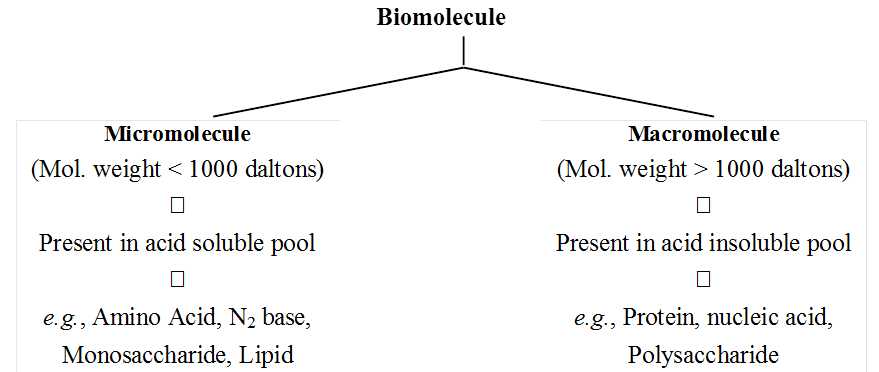
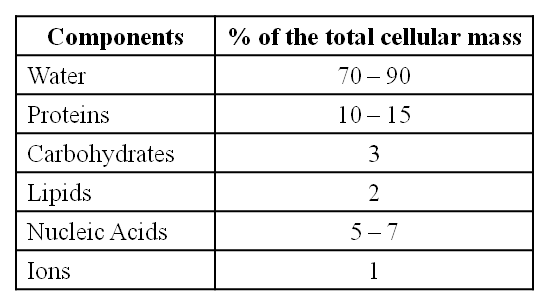
(ii) The most abundant element in cell/living matter is oxygen. O > C > N > H
(iii) Fe++ and Cu++ are found in cytochromes.
(iv) The concentration of the cations inside the cell is K > Na > Ca.
How To Analyse Chemical Composition?
In order to study the various biomolecules found in living tissues (a vegetable or a piece of liver etc.), the tissue is ground in trichloroacetic acid (Cl3CCOOH) using pestle and mortar.
The resultant slurry is strained through cheese cloth or cotton and we obtain two fractions.
The filtrate is called acid soluble pool while the retentate is called acid insoluble fraction.
The acid soluble pool represents roughly the cytoplasmic composition.
The macro molecules from cytoplasm and organelles become the acid-insoluble fraction.
Chemicals present in both the fractions are further separated by various analytical techniques and identified.
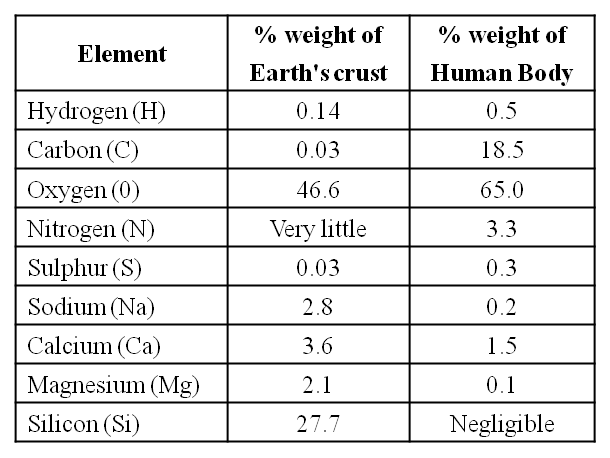
Average Composition of Cells
Note : Protein > Nucleic acid > Carbohydrates > Lipids
The acid soluble pool contains chemicals called biomicromolecules as they have small molecular mass of 18-800 daltons approximately.
The acid insoluble fraction contains chemicals with large molecular mass of more than 800 daltons, they are biomacromolecules.
Biomacromolcules are large size, high molecular weight, complex molecules that are formed by condensation of biomicromolecules.
Their molecular mass is in the range of ten thousand daltons and above.
Biomacromolecules are of three types-proteins, nucleic acids and polysaccharides.
Note : Though lipids have a molecular mass similar to that of micromolecules i.e. less than 800 Da, but they do not appear in the acid Soluble pool due to their non-polar nature.
All biomacromolecules are polymers except lipids.
Polymers are formed by process of union of repeating subunits, each subunit being called monomer.
Monomers are simple small sized low molecular weight molecules which cannot be hydrolysed further into smaller subunits.
Polymers occur in the form of threads.
They are folded variously to form three-dimensional shapes required for their functioning.
A list of Representative Inorganic Constituents of Living Tissues

Depending upon the molecular weight and solubility, biomolecules are divided into two categories.
(a) Micromolecules are small sized, have low molecular weight, simple molecular structure and high solubility in the intracellular fluid matrix. These include water, mineralrs, gases, carbohydrates, lipids, amino acids and nucleotides.
(b) Macromolecules are large sized, have larger molecular weight, complex conformation and low solubility in the intracellular fluid matrix. They are generally formed by polymerisation of micromolecules. These include polysaccharides, proteins and nucleic acids.
Analytical techniques, when applied to the compound give us an idea of the molecular formula and the probable structure of the compound.
All the carbon compounds that we get from living tissue can be called "Biomolecules".
However, living organisms also have inorganic elements and compounds.
When the tissue is fully burnt all carbon compounds are oxidised to gaseous form (CO2, water vapour) and are removed.
What is remaining is called "Ash".
This ash contains inorganic elements (like calcium, magnesium etc.).
Inorganic compounds like sulphate, phosphate etc. are also seen in the acid soluble fraction.
Therefore, elemental analysis gives elemental composition of living tissues in the form of H, O, Cl, C etc. while analysis of compounds gives an idea of the kind of organic and inorganic constituents present in living tissues.
From a chemistry point of view, one can identify functional groups like aldehydes, ketones, aromatic compounds etc.
But from a biological point of view, we shall classify them into aminoacids, nucleotide bases, fatty acids etc.
Chapter 9
Biomolecules
The biosphere has a vast variety of living species. Carbon, hydrogen, oxygen, and numerous other elements, as well as their respective contents, are acquired per unit mass of living tissue when an elemental analysis is done on plant tissue, animal tissue, or microbial paste. A similar list of elements will be obtained if the same analysis is performed on a piece of the earth's crust as an example of non-living stuff. A sample of live tissue contains all of the elements found in a sample of the earth's crust. However, a closer look reveals that any living organism has a higher relative quantity of carbon and hydrogen in relation to other elements than the earth's crust.
How to Analyse Chemical Composition?
A natural substance's elemental analysis reveals that it is made up of several elements such as carbon, hydrogen, oxygen, chlorine, and so on. Analytical procedures provide information on various organic and inorganic compounds, as well as their molecular formulas and structures. They also aid in the separation and purification of one component from another. Simple experiments can be used to determine the chemical composition of biomolecules. Crush and combine a piece of living tissue with an acid. We get two pieces after filtering it. The acid-soluble fraction of the filtrate is retained on the filter membrane, while the acid-insoluble fraction is retained on the filter membrane. This indicates that there are two or more substances with distinct characteristics within the tissues.Thousands of organic molecules have been discovered in the acid-soluble pool by scientists.

Constituents of Living Tissues
Take another piece and burn it till all the moisture in it is evaporated. When carbon compounds are burned, they are all oxidized. Inorganic substances such as calcium, magnesium, sulfate, phosphate, and others are formed in the tissue by the ash that has been left out.
Analytical techniques, when applied to a chemical, provide us with an estimate of its molecular formula and likely structure. 'Biomolecules' refers to all carbon compounds obtained from living tissues. All carbon-containing chemicals (organic compounds) found in living organisms are classified as biomolecules. They are organic substances found in living cells that have a role in the organism's maintenance and metabolic activities. Inorganic elements and compounds, on the other hand, are found in living beings. As a result, elemental analysis provides information on the composition of live tissues in terms of hydrogen, oxygen, chlorine, carbon, and other elements, whereas compound analysis provides information on the types of organic and inorganic constituents found in living tissues.
Functional groups such as aldehydes, ketones, aromatic molecules, and others can be detected in living tissues from a chemical standpoint. However, amino acids, nucleotide bases, fatty acids, and carbohydrates make up living tissues from a biological standpoint.
Amino acids are chemical molecules that have both an amino and an acidic group as substituents on the same carbon, the -carbon. As a result, they are known as -amino acids. They're methanes that have been replaced. The four valency locations are occupied by four substituent groups. Hydrogen, carboxyl group, amino group, and a variable group known as R group are the four groups. There are numerous amino acids in the R group due to its nature. However, there are only twenty varieties of those found in proteins. The R group in these proteinaceous amino acids could be a hydrogen (glycine), a methyl group (alanine), hydroxy methyl (serine), or something else entirely. The amino, carboxyl, and R functional groups make up the chemical and physical properties of amino acids.Acidic (e.g., glutamic acid), basic (lysine), and neutral (valine) amino acids are classified by the number of amino and carboxyl groups they contain.
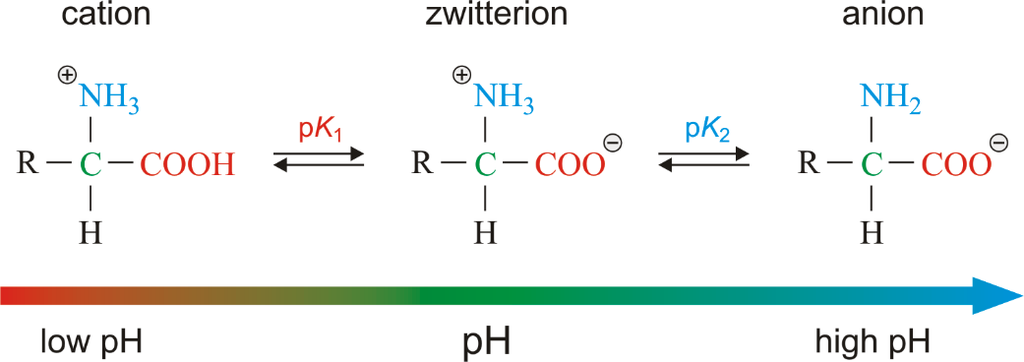
Similarly, aromatic amino acids exist (tyrosine, phenylalanine, tryptophan). The ionizability of the –NH2 and –COOH groups of amino acids is a unique feature. As a result, the structure of amino acids alters in different pH solutions.
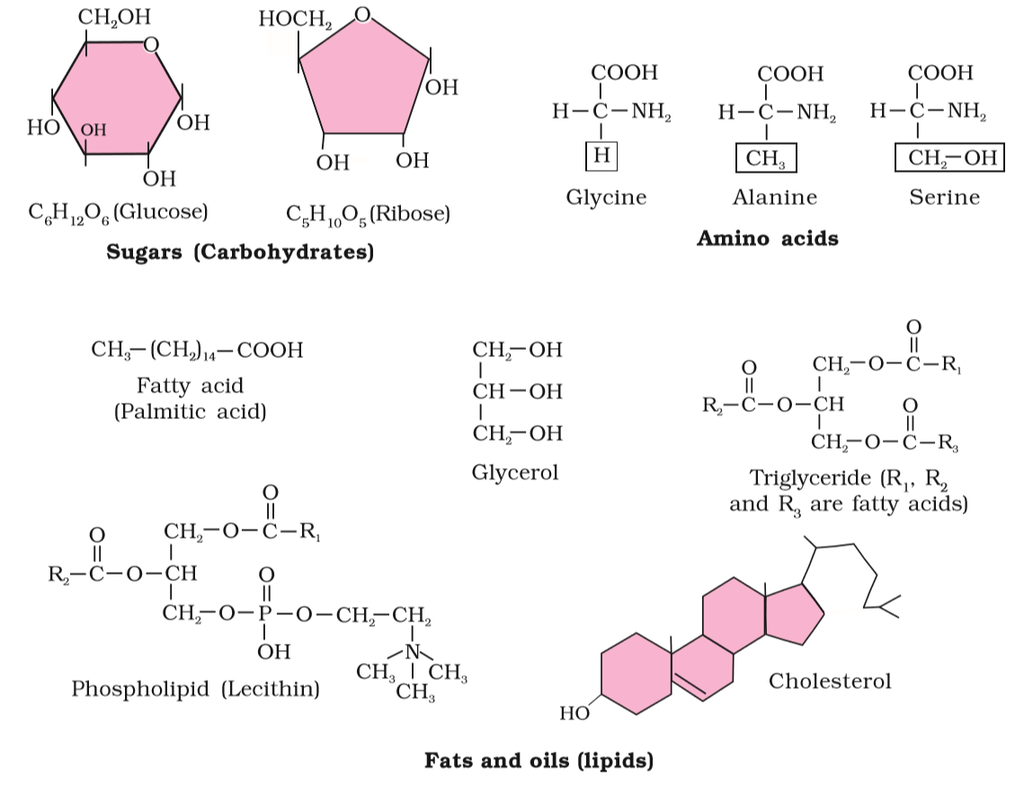
Figure 2 (a): Diagrammatic representation of small molecular weight organic compounds in living tissues
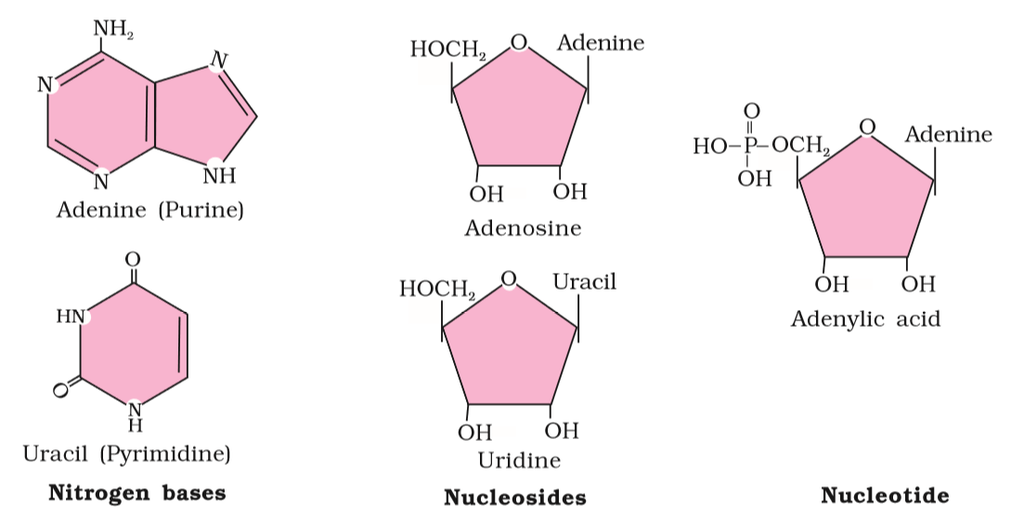
Figure 2(b): Diagrammatic representation of small molecular weight organic compounds in living tissues
Heterocyclic rings can be found in a variety of carbon compounds found in living beings. Adenine, guanine, cytosine, uracil, and thymine are examples of nitrogen bases. They're called nucleosides when they're found linked to sugar. Nucleotides are formed when a phosphate group is additionally esterified to the sugar. Nucleosides include adenosine, guanosine, thymidine, uridine, and cytidine. Nucleotides include adenylic acid, thymidylic acid, guanylic acid, uridylic acid, and cytidylic acid. Only nucleotides make up nucleic acids like DNA and RNA. DNA and RNA are two types of genetic material.

 ACME SMART PUBLICATION
ACME SMART PUBLICATION
